The Biote lawsuit is not a single case — it’s a series of high-stakes legal battles that have surrounded the hormone therapy company since 2022. The most prominent disputes involve allegations that company executives hijacked the firm from its founder through a deeply controversial SPAC merger, enriching themselves while diluting shareholder value. Separate from those shareholder disputes, Biote also faces product liability suits from patients who say its hormone pellets caused serious medical harm. Together, these cases have resulted in settlements exceeding $136 million, with additional litigation still ongoing in 2026. Facial abuse lawsuit
This guide covers every major Biote lawsuit — who sued, what they alleged, how much was settled, and what’s still unresolved. If you’re a former patient, investor, or shareholder, this page will help you understand where things stand and whether you may have your own legal options. Carnival Triumph Lawsuit
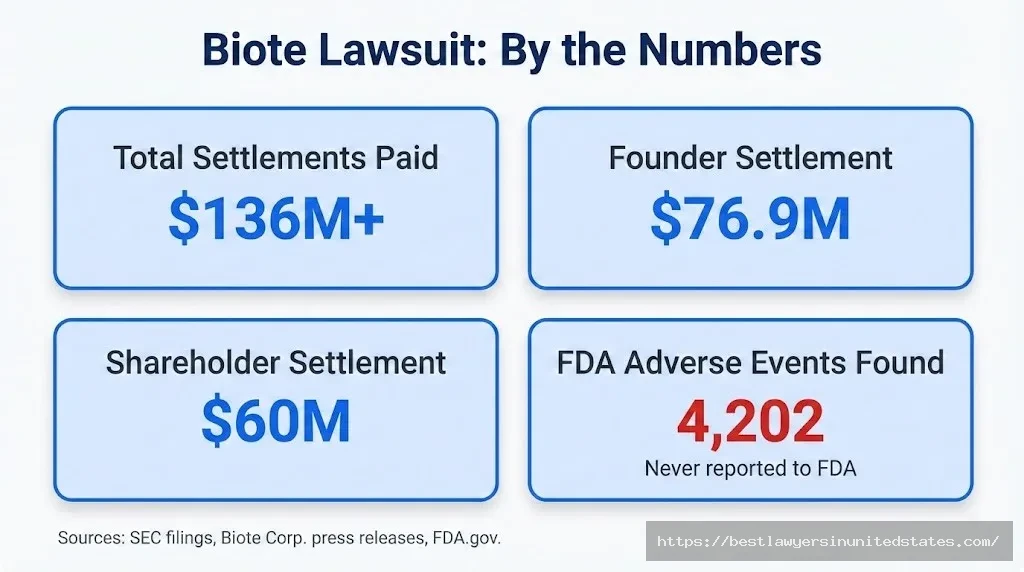
Quick Summary Biote Corp. (NASDAQ: BTMD) has faced multiple lawsuits since going public via a SPAC in May 2022. Founder Dr. Gary Donovitz settled claims for approximately $76.9 million (with a final $18.5M payment made in January 2026). Shareholder Marci Donovitz settled for $60 million. A third shareholder lawsuit filed in 2024 remains active. Separately, patient lawsuits allege serious injuries from Biote’s compounded hormone pellets, and the FDA previously found over 4,200 unreported adverse events tied to Biote products.
Biote Lawsuit At a Glance
| Detail | Information |
|---|---|
| Company | Biote Corp. (NASDAQ: BTMD), Irving, Texas |
| Founded by | Dr. Gary S. Donovitz (2012) |
| Business | Hormone optimization / bioidentical hormone replacement therapy (BHRT) via pellets |
| Went public | May 26, 2022 via SPAC merger with Haymaker Acquisition Corp. III |
| Primary lawsuit type | Shareholder / breach of fiduciary duty (SPAC fraud claims) |
| Secondary lawsuit type | Patient product liability (hormone pellet injuries) |
| Total settlements paid (approx.) | >$136 million |
| Case status (2026) | Founder/Marci lawsuits settled; Miller Trust lawsuit ongoing; patient cases ongoing |
| Key courts | Delaware Court of Chancery; 101st Judicial District Court, Dallas County, TX |
What Is the Biote Lawsuit About?
Background: A Hormone Therapy Company Goes Public — and Into Court
Biote (originally BioTE Medical) was built by Dr. Gary Donovitz into a leading provider of pellet-based bioidentical hormone replacement therapy. The business trained medical practitioners to administer subcutaneous hormone pellets, offering a recurring revenue model tied to patient re-pelleting cycles. By 2021, it was a profitable, growing company — and a target for a SPAC merger.
In December 2021, Biote signed a Business Combination Agreement (BCA) with Haymaker Acquisition Corp. III, a blank-check company. The deal closed on May 26, 2022, and Biote began trading on NASDAQ under the ticker BTMD. But almost immediately, the story turned legal. Dr. Donovitz — the man who built the company — alleged he had been deceived into signing away key contractual rights, and that the SPAC deal was structured to funnel tens of millions of dollars to executives and insiders at his and other shareholders’ expense.
What Is a SPAC, and Why Does It Matter Here?
A SPAC (Special Purpose Acquisition Company) is a shell company that raises money through an IPO, then uses those funds to merge with a private company — effectively taking it public. The SPAC’s investors can choose to redeem their shares for cash rather than hold stock in the merged company. In Biote’s case, the allegations center on the claim that executives knew investor redemptions would be catastrophically high — draining virtually all the cash from the merger — but pushed the deal forward anyway because doing so personally rewarded them with stock, management control, and financial benefits they hadn’t earned.
According to court filings, over $70 million in merger consideration was allegedly diverted to Biote insiders, while the merger itself provided almost no usable cash to the company. Shareholders who had trusted executives to act in their best interests claim they were left holding devalued stock.
Timeline of Key Events
| Date | Event | Details |
|---|---|---|
| 2012 | Biote founded | Dr. Gary Donovitz founds BioTE Medical as a hormone pellet training and marketing company |
| Dec. 13, 2021 | SPAC agreement signed | Biote signs Business Combination Agreement with Haymaker Acquisition Corp. III |
| May 26, 2022 | SPAC merger closes; Biote goes public | BTMD begins trading on NASDAQ; massive redemptions leave little cash |
| June 22, 2022 | Donovitz files first Texas lawsuit | Dr. Donovitz sues Cooley LLP, SPAC promoters — alleging fraudulent inducement of BCA and FAA |
| 2022–2023 | BioTE Medical countersuit & attorney fee trial | Biote sues Donovitz for breach of Founder Advisory Agreement; Dallas jury awards Biote $4.67M in attorney fees |
| Feb. 2024 | Settlement term sheet announced | Biote announces agreement to repurchase all of Donovitz’s shares for ~$76.9M over 3 years |
| April 29, 2024 | Definitive settlement finalized | Formal settlement agreement signed; Donovitz gets mutual release, Biote gets 18.4M shares back |
| June 5, 2024 | Marci Donovitz files Delaware lawsuit | Ex-wife/trust trustee sues CEO Weber, Chairman Beer, Haymaker, and Cooley LLP for breach of fiduciary duty |
| July 3, 2024 | Marci Donovitz settles for $60M | Biote agrees to repurchase ~8.3 million shares; settlement resolves all related claims |
| July 16, 2024 | Third lawsuit filed (Miller Trusts) | Brewer law firm files on behalf of Russell and Mary Miller’s trusts — same SPAC fraud allegations, ongoing |
| Oct. 7, 2025 | Biote sues in Delaware to block indemnification | Biote files for injunction barring Donovitz from pursuing Texas indemnification cross-claims predating the April 2024 settlement |
| Nov. 2025 | Settlement amended | Biote and Donovitz amend their deal; Biote agrees to pay $18.5M in January 2026 to fully close out remaining payment obligations |
| Jan. 2026 | Final Donovitz payment made | Biote pays the $18.5M final installment, fully settling the multi-year Donovitz litigation |
The Three Major Shareholder Lawsuits Explained
Lawsuit #1: Dr. Gary Donovitz v. Biote
SETTLED
~$76.9 Million
Filed: June 2022 | Settled: April 2024, fully paid January 2026
Dr. Donovitz, who built Biote from the ground up starting in 2012, alleged that the executives he hired — particularly CEO Teresa Weber and General Counsel Mary Elizabeth Conlon — deceived him into signing the SPAC merger agreements. He claimed his contracted rights were altered in the final electronic version of the Founder Advisory Agreement (FAA) without his knowledge, that key financial protections like a minimum cash closing condition were stripped from deal documents, and that the SPAC transaction ultimately diluted his ownership stake by nearly $220 million.
He also alleged that Conlon — who had previously served as his personal attorney for nearly a decade — betrayed his trust by acting in the interests of the company’s new leadership. The case was initially filed in Texas, and a parallel contract suit filed by BioTE Medical against Donovitz resulted in a Dallas jury awarding Biote $4.67 million in attorney fees after Donovitz stipulated to liability on breach-of-contract claims before trial.
The resolution: Biote agreed to repurchase all 18.4 million of Donovitz’s shares (approximately 5.1 million Class A Shares and 13.3 million Paired Interests) for a total of approximately $76.9 million, paid over three years at an average of $4.17 per share. In November 2025, the parties amended the deal so Biote could pay $18.5 million as a final lump sum in January 2026, fully closing the matter.
Lawsuit #2: Marci Donovitz (Donovitz Family Irrevocable Trust) v. Biote
SETTLED
$60 Million
Filed: June 5, 2024 | Settled: July 3, 2024 (remarkably fast — within weeks)
Marci Donovitz, the ex-wife of Dr. Gary Donovitz and trustee of the Donovitz Family Irrevocable Trust, filed suit in the Delaware Court of Chancery against Biote CEO Teresa Weber, Executive Chairman Marc Beer, General Counsel Mary Elizabeth Conlon, Haymaker Sponsor III LLC, Steven J. Heyer, and law firm Cooley LLP.
Her lawsuit alleged that these defendants knew the SPAC deal would result in “astronomical redemptions” that would drain almost all the cash raised, yet pushed it forward anyway because doing so rewarded insiders with stock, cash, and management control worth approximately $135 million. Marci was allegedly misled about the likely impact of the merger on her shares’ value, and critical risk information was withheld from her as a beneficial shareholder.
The settlement required Biote to repurchase all of Marci Donovitz’s approximately 8.3 million shares. Her attorney, William Brewer III, called the outcome a validation of their claim that the transaction was a scheme to enrich company insiders at shareholders’ expense.
Lawsuit #3: Miller Trust v. Biote (and Related Parties)
ONGOING
Damages Sought: Disgorgement of ~$70M+
Filed: July 16, 2024 | Status: Active as of early 2026
Filed by Brewer, Attorneys & Counselors on behalf of Russell and Mary Miller, co-trustees of the Yosaki and Mioko Trusts, this third lawsuit raises essentially the same core allegations as the Marci Donovitz case: that Biote executives conspired to close a destructive SPAC deal that diluted shareholders’ value while enriching insiders. Defendants named include CEO Teresa Weber, Executive Chairman Marc Beer, Mary Elizabeth Conlon, Haymaker Sponsor III LLC, Steven J. Heyer, and Cooley LLP.
The complaint accuses the insider defendants of conspiring to divert approximately $70 million in merger consideration to themselves and gain control of an enterprise they didn’t build, and asks the court to order disgorgement of those gains. This case remains in active litigation as of early 2026.
The Core SPAC Fraud Allegations Against Biote
Across all three shareholder lawsuits, the following claims appear repeatedly in court documents:
- Executives knew for months before closing that “astronomical redemptions” by Haymaker shareholders would drain virtually all of the SPAC’s cash — yet pushed the deal through anyway.
- Approximately $70 million in merger consideration was improperly diverted to Biote insiders, and approximately $135 million in total cash and stock flowed to all defendants combined.
- The minimum cash closing condition — a financial protection that would have blocked the merger if Haymaker had insufficient cash to pay Donovitz — was removed from deal documents just before the FAA was finalized.
- Shareholders were misled about the risks of the transaction and not told that high redemptions had already materialized, making the deal value-destructive before it even closed.
- Cooley LLP, as outside counsel, allegedly aided and abetted breaches of fiduciary duty by the named executives.
Settlements: How Much Did Biote Pay and to Whom?
Settlement Fund Breakdown
| Lawsuit | Settlement Amount | Form of Payment | Status |
|---|---|---|---|
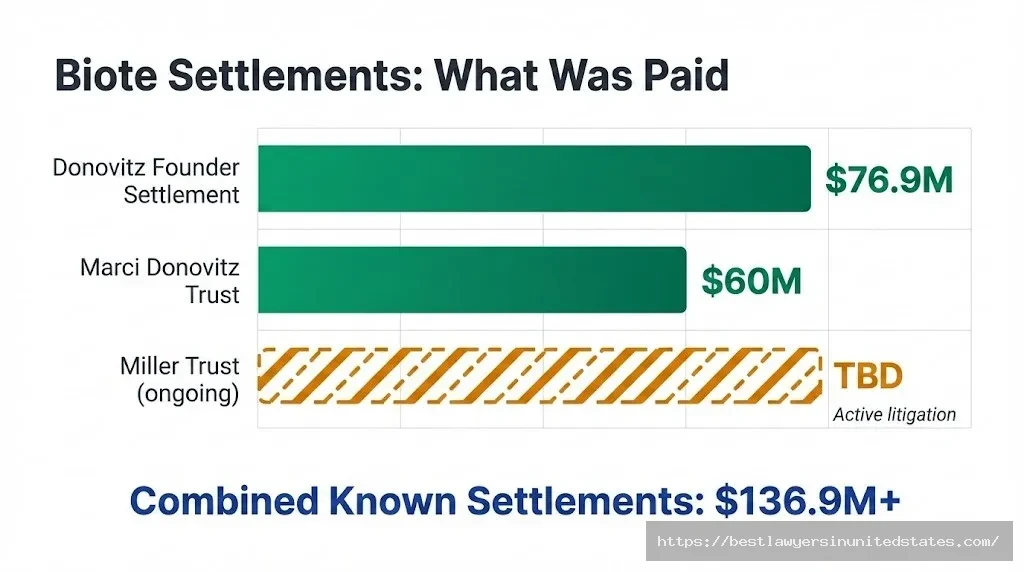
| Dr. Gary Donovitz v. Biote | ~$76.9 million | Share repurchase over 3 years ($30M at closing, then installments); final $18.5M paid Jan. 2026 | Fully Paid |
| Marci Donovitz (Trust) v. Biote | $60 million | Share repurchase (8.3M shares) | Settled July 2024 |
| Miller Trust v. Biote et al. | TBD | Ongoing — no settlement announced | Ongoing |
| Patient product liability cases | TBD (individual cases) | Case-by-case; no class settlement known | Ongoing |
| TOTAL PAID (known) | >$136.9 million | — | — |
Important Note on These Settlements These are not consumer class action settlements where ordinary customers can file claims for cash. The Donovitz and Marci Donovitz settlements are private corporate resolutions between Biote and specific shareholders. If you’re a patient who experienced adverse effects from Biote hormone pellets, you would need to consult a personal injury or medical malpractice attorney about your individual situation. Endurance Warranty Lawsuit
The Patient Lawsuits: Hormone Pellet Injuries
What Patients Are Alleging
Separate from the corporate governance battles, Biote faces product liability suits from patients who claim its compounded hormone pellets caused serious physical harm. One of the most prominent cases was filed in West Virginia by plaintiff Timothy Koontz, who alleged that Biote’s testosterone pellets led to blood clotting, abdominal vein thrombosis, and multiple life-saving surgeries.
The core complaint in patient suits is that Biote’s proprietary dosing algorithm routinely produced testosterone levels far above physiologically normal ranges — so-called “supraphysiologic” dosing — which plaintiffs allege increases the risk of serious cardiovascular and other complications. Patients say they weren’t adequately warned about these risks when they chose pellet therapy.
The FDA Connection: 4,202 Unreported Adverse Events
The patient lawsuits gain additional weight from a federal regulatory finding. During an inspection of BioTE Medical in 2018, FDA investigators discovered 4,202 adverse events associated with compounded hormone pellets that had never been reported to the agency. The FDA published a statement noting that the possibly associated events included endometrial cancer, prostate cancer, strokes, heart attacks, deep vein thrombosis, cellulitis, and pellet extrusion.
The FDA’s statement noted that BioTE had an online portal to collect adverse event data from its practitioners — but never reported that information to the FDA, nor passed it along to the outsourcing facilities that actually manufactured the pellets. This failure to report spanned five years, from 2013 to 2018. The FDA statement said the agency was continuing to investigate the matter.
⚠️ If You Were a Biote Patient Who Experienced Adverse Effects If you received Biote hormone pellet therapy and experienced serious side effects — including blood clots, cardiovascular events, unusual cancer diagnoses, pellet extrusion, or other complications — you may have legal options. These are individual cases, not a class action settlement. Contact a personal injury or medical malpractice attorney to discuss your situation. Many offer free initial consultations.
What Biote Says
Biote has denied wrongdoing in the patient lawsuits and maintains that its hormone therapy products comply with applicable safety and regulatory standards. The company has emphasized that it provides practitioners with detailed training and that the pellets are manufactured by FDA-regulated 503B outsourcing facilities. Following the adverse event reporting controversy, the company revised some of its practices around patient safety data.
Comparison: Biote Patient Lawsuits vs. Similar Hormone Therapy Cases
| Case/Company | Core Allegation | Type | Status |
|---|---|---|---|
| Biote / BioTE Medical (patient suits) | Supraphysiologic dosing; failure to warn; unreported adverse events | Product liability / medical malpractice | Ongoing |
| Wyeth (Prempro) HRT MDL | Estrogen-progestin therapy linked to breast cancer; failure to warn | Mass tort / class action | Largely resolved (thousands of individual settlements) |
| AbbVie (AndroGel) | Testosterone therapy heart attack / stroke risk not disclosed | Multi-district litigation | Largely resolved |
| EvexiPEL (competitor pellet company) | Similar pellet overdosing/dosing algorithm concerns | Individual suits | Mixed outcomes |
Current Status & Latest Updates (2026)
What’s Resolved
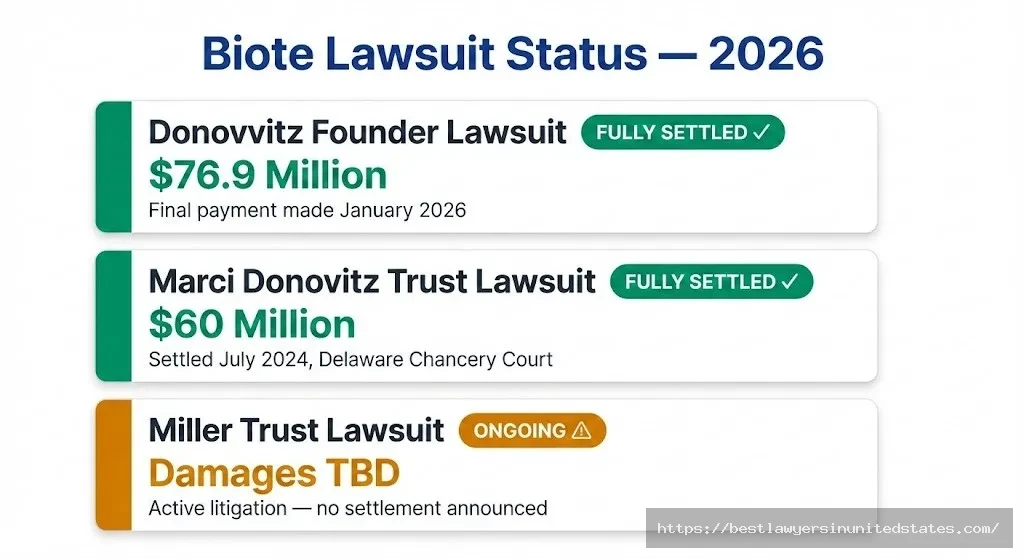
The Donovitz founder litigation is fully resolved. As of January 2026, Biote completed its final $18.5 million payment to Dr. Donovitz under the amended settlement agreement, fully satisfying the multi-year share repurchase obligation. The Marci Donovitz settlement, reached in July 2024, is also closed. All related claims between those parties have been mutually released.
What’s Still Active
Two significant areas of litigation remain live. The Miller Trust lawsuit — the third shareholder case alleging the same SPAC insider enrichment scheme — is ongoing and has not settled. And in October 2025, Biote itself went back to the Delaware Court of Chancery, filing for an injunction to block Dr. Donovitz from pursuing a Texas court cross-claim seeking indemnification for liability that pre-dated the April 2024 settlement. Biote argues the original settlement agreement prohibits such claims; Donovitz apparently disagrees. That dispute was active as of late 2025.
On the patient side, individual product liability cases involving Biote hormone pellets continue to move through the court system. No class-wide settlement has been announced.
Biote’s Business Position in 2026
Despite the legal turbulence, Biote continues operating. The company reported Q3 2025 net income of $9.2 million, though operating income declined 32.1% year-over-year. Adjusted EBITDA was $12.9 million, down 20.5% from the prior-year quarter. The company has undergone organizational restructuring and changed its CEO — Bret Christensen now leads the company, following the departure of Teresa Weber, who was named as a defendant in multiple lawsuits. Biote continues to train practitioners in hormone optimization and generate revenue from its pellet therapy business model.
Upcoming Dates to Watch
| Matter | Status / Timeframe | What to Watch For |
|---|---|---|
| Dr. Gary Donovitz litigation | Fully settled, Jan. 2026 | Watch Delaware injunction dispute over indemnification claims |
| Marci Donovitz settlement | Fully closed, 2024 | No further developments expected |
| Miller Trust lawsuit | Active litigation | Any trial date announcement, pretrial rulings, or settlement news |
| Delaware indemnification injunction | Filed Oct. 2025; ongoing | Court ruling on whether Donovitz’s Texas indemnification claim is barred by 2024 settlement |
| Patient product liability cases | Multiple, ongoing | Any bellwether trial results or class certification attempts |
| FDA investigation (adverse event reporting) | Opened 2018; status unclear | Any enforcement action or resolution from FDA |
Who Can Sue Biote? Do You Have a Claim?
If You Were a Biote Shareholder Before or During the 2022 SPAC Merger
If you held shares in BioTE Holdings, LLC or received equity as part of the SPAC transaction and believe you suffered losses due to insider misconduct or breach of fiduciary duty, you may have grounds for a legal claim. The Miller Trust case demonstrates that additional shareholder plaintiffs are still actively pursuing these allegations. Contact a securities litigation attorney — most offer free consultations — to evaluate your specific situation.
Quick Answer: Shareholder Eligibility Former BioTE Holdings, LLC shareholders and those who received equity through the 2022 SPAC merger may have viable claims. Two shareholders (the Donovitzes) settled for a combined $136+ million. A third case is active. You’d need to have held equity in the relevant entities and demonstrate harm from the alleged insider conduct.
If You Were a Patient Who Experienced Adverse Effects
Biote’s pellet therapy has been associated — through FDA findings and individual lawsuits — with serious medical complications including blood clots, DVT, cardiovascular events, cancer, and pellet extrusion. If you received Biote hormone pellets and subsequently experienced such complications, you may have a product liability or medical malpractice claim against Biote, the outsourcing facility that made your pellets, and/or the practitioner who administered them.
- You received compounded hormone pellets through a Biote-certified practitioner
- You experienced a serious medical complication — blood clot, DVT, stroke, heart attack, cancer, extrusion, or other significant health event
- There’s a plausible link between the pellet therapy and your injury (timing, lab results, medical records)
- You are within your state’s statute of limitations for personal injury (typically 2–3 years, varies by state)
Who Likely Does NOT Have a Claim
- ❌ Patients who had Biote pellets with no adverse outcomes
- ❌ Anyone who signed a valid arbitration agreement with their Biote-certified provider
- ❌ People whose claims are barred by their state’s statute of limitations
- ❌ Those who received FDA-approved testosterone pellets (Testopel), not Biote’s compounded pellets
Required Documentation If You’re Pursuing a Claim
| Document | Why You Need It | Where to Find It |
|---|---|---|
| Medical records / treatment notes | Establishes you received Biote pellet therapy and timing | Your practitioner’s clinic; request via patient portal or written request |
| Lab results (hormone panels) | May show supraphysiologic testosterone levels | Same as above; blood draw labs |
| Records of adverse event | Establishes the harm and its timing relative to treatment | Hospital records, specialist records, emergency care |
| Pharmacy/pellet records | Identifies the outsourcing facility that compounded your pellets | Your practitioner; compounding pharmacy |
| Any informed consent forms you signed | Important for evaluating what risks you were or weren’t warned about | Your practitioner’s office |
| Billing/payment records | Confirms dates of treatment | Your EOB from insurer or direct payment receipts |
Do You Need a Lawyer?
Key Point Unlike consumer class action settlements — where you simply fill out an online claim form — the Biote lawsuits involve complex corporate, securities, and medical liability issues. Whether you’re a shareholder or a patient, an attorney consultation is strongly recommended before taking action.
For Shareholders
Securities and shareholder litigation is specialized. The Miller Trust case is actively being litigated by Brewer, Attorneys & Counselors, a Texas firm that has handled all three major Biote shareholder cases. If you believe you have a related claim as a BioTE Holdings equity holder, reaching out to a firm experienced in shareholder litigation and SPAC disputes would be a logical first step. Most securities litigation attorneys work on contingency — you pay nothing upfront and they only collect if you win.
For Patients
Product liability and medical malpractice cases are also complex and time-sensitive. Statutes of limitations vary by state, typically running 2–3 years from the date of injury or discovery of harm. If you were harmed by Biote pellet therapy, don’t delay in seeking a legal consultation. Many personal injury attorneys offer free case evaluations and work on contingency.
For attorney referrals, you can contact: admin@bestlawyersinunitedstates.com
Frequently Asked Questions
What is the Biote lawsuit about?
Quick Answer: There are multiple Biote lawsuits — the most prominent ones involve shareholders claiming executives fraudulently enriched themselves through the 2022 SPAC merger; separate lawsuits involve patients claiming serious injuries from Biote hormone pellets.
The shareholder cases center on allegations that Biote CEO Teresa Weber, Executive Chairman Marc Beer, and others orchestrated a SPAC merger that diverted over $70 million in merger consideration to insiders while leaving existing shareholders with devalued stock. The patient cases allege that Biote’s compounded testosterone and estrogen pellets are dosed at dangerously high levels and that Biote failed to disclose or properly report thousands of adverse events.
Who is Dr. Gary Donovitz and why did he sue Biote?
Quick Answer: Dr. Donovitz founded Biote in 2012 and sued the company he built after alleging that executives he hired deceived him into a fraudulent SPAC deal that diluted his ownership by ~$220 million.
He alleged the final version of his Founder Advisory Agreement was altered without his knowledge, that financial protections were stripped from deal documents, and that his personal attorney (Conlon) betrayed his trust by acting against his interests. He ultimately settled for approximately $76.9 million in share repurchases, fully paid in January 2026.
How much did Biote pay to settle the lawsuits?
Quick Answer: Known settlements total over $136 million — approximately $76.9M to Dr. Gary Donovitz and $60M to Marci Donovitz. Additional litigation (Miller Trust, patient cases) remains ongoing.
Is there a class action settlement where patients can file claims?
Quick Answer: Not as of early 2026. There is no announced consumer class action settlement for Biote patients to file claims against. Patient cases are being pursued individually.
If you were harmed by Biote’s hormone pellets, you would need to pursue your own individual legal claim rather than filing under a class settlement. Contact a personal injury attorney to evaluate your options.
What were the FDA findings about Biote adverse events?
Quick Answer: During a 2018 inspection, the FDA found 4,202 adverse events associated with BioTE Medical’s compounded hormone pellets that had never been reported to the agency over a five-year period.
Possibly associated adverse events included endometrial cancer, prostate cancer, strokes, heart attacks, deep vein thrombosis, cellulitis, and pellet extrusion. The FDA noted that BioTE collected this data internally but never forwarded it to regulators or to the outsourcing facilities that manufactured the pellets. The FDA investigation was described as ongoing at the time of that public statement.
Who were the defendants in the shareholder lawsuits?
Quick Answer: The primary defendants were Biote CEO Teresa Weber, Executive Chairman Marc Beer, General Counsel Mary Elizabeth Conlon, Haymaker Sponsor III LLC, Steven J. Heyer, and law firm Cooley LLP.
What happened to Teresa Weber, Biote’s CEO?
Quick Answer: Teresa Weber, who served as Biote’s CEO and was named as a defendant in multiple lawsuits, is no longer in the role. Bret Christensen became CEO and was leading the company as of late 2025.
Is Biote still in business?
Quick Answer: Yes. Biote (NASDAQ: BTMD) continues to operate as a hormone optimization company, training practitioners and selling pellet therapy programs. The company reported positive net income in Q3 2025.
Despite the legal battles and management changes, Biote’s core business — licensing its method to medical practitioners and selling ancillary products and nutraceuticals — remains active. Its stock trades on NASDAQ under the ticker BTMD.
What is a SPAC and why is it central to the Biote lawsuits?
Quick Answer: A SPAC (Special Purpose Acquisition Company) is a shell company that raises cash via IPO then merges with a private company to take it public. Biote’s 2022 SPAC merger is alleged to have been structured to enrich insiders at the expense of existing shareholders.
SPAC investors can redeem their shares for cash rather than hold merged-company stock. In Biote’s case, redemptions were allegedly so high that almost no cash remained after the merger — yet executives proceeded with the deal and personally received significant compensation and equity, which is the heart of the fraud allegations.
Can I still sue Biote as a patient who was harmed?
Quick Answer: Possibly — but statutes of limitations apply. Generally, you have 2–3 years from the date of your injury or the date you discovered (or should have discovered) the harm to file a claim, depending on your state.
Product liability and medical malpractice cases are time-sensitive. If you experienced a serious adverse event following Biote hormone pellet therapy, consult a personal injury attorney as soon as possible to preserve your legal options.
Were Biote’s pellets FDA-approved?
Quick Answer: The individual hormones (testosterone, estradiol) are FDA-approved substances, but Biote’s compounded pellets themselves are not FDA-approved drug products.
Biote’s pellets are custom-compounded by 503B outsourcing facilities. While these facilities are regulated by the FDA, the specific compounded pellet products are not subject to the same pre-market approval process as brand-name pharmaceutical drugs. This distinction is central to both the patient lawsuits and the FDA adverse event reporting controversy.
What is the Miller Trust lawsuit about, and is it still ongoing?
Quick Answer: The Miller Trust lawsuit, filed July 2024, raises the same SPAC insider enrichment allegations as the Donovitz cases and remains active in litigation as of early 2026.
Filed by Brewer, Attorneys & Counselors on behalf of Russell and Mary Miller’s trusts, this case names the same defendants and seeks disgorgement of profits improperly diverted from shareholders. No settlement has been announced.
What happens to Cooley LLP, the law firm named as a defendant?
Quick Answer: Cooley LLP was named as a defendant in the Marci Donovitz and Miller Trust lawsuits, accused of aiding and abetting breaches of fiduciary duty. Cooley’s liability in the settled Marci case was resolved as part of that settlement; the Miller Trust case against Cooley remains active.
What are supraphysiologic hormone doses and why do they matter legally?
Quick Answer: Supraphysiologic doses are hormone levels that significantly exceed the natural physiological range. Patient lawsuits claim Biote’s dosing algorithm routinely produced these levels, creating health risks that patients weren’t warned about.
Standard hormone replacement therapy aims to restore hormone levels to a normal physiological range. Critics of Biote’s approach — and some patient plaintiffs — allege the company’s proprietary dosing methodology pushed levels far beyond that range, increasing risks of blood clots, cardiovascular events, and other serious conditions. Biote denies these characterizations.
Where can I find the official court documents for the Biote lawsuits?
Quick Answer: The primary shareholder cases were filed in the Delaware Court of Chancery and the 101st Judicial District Court of Dallas County, Texas. SEC filings by Biote Corp. (BTMD) also contain significant settlement-related disclosures.
Court documents can be searched via the Delaware Court of Chancery’s eCourts system or Dallas County’s district court records. Settlement agreements filed with the SEC are available through the EDGAR database at sec.gov. Search for “biote” or ticker “BTMD.” Are Lawsuit Settlements Taxable?
Key Takeaways
The Biote lawsuit saga is a cautionary tale about SPAC-era corporate governance and the risks of compounded hormone therapies. Here’s the essential summary going into 2026:
- The founder who built Biote, Dr. Gary Donovitz, settled claims for ~$76.9 million — fully paid January 2026.
- His ex-wife and trust trustee, Marci Donovitz, settled separately for $60 million just weeks after filing suit.
- A third shareholder lawsuit (Miller Trust) is actively litigating the same core SPAC fraud claims.
- Patient injury lawsuits allege serious harm from high-dose hormone pellets; no class settlement exists.
- The FDA found 4,202 unreported adverse events tied to BioTE Medical’s products during a 2018 inspection.
- Biote continues to operate but has undergone significant management changes, including a new CEO.
Need Legal Help? If you’re a former Biote shareholder or a patient who suffered harm from Biote hormone therapy, your situation deserves a proper legal evaluation. Contact a qualified attorney who handles securities litigation or personal injury/medical malpractice. Many offer free consultations. You can also reach out to admin@bestlawyersinunitedstates.com for a referral.
Disclaimer: This article is for informational purposes only and does not constitute legal advice. Claude is not a lawyer. Settlement amounts and case statuses are based on publicly available sources as of February 2026. Always consult a licensed attorney for advice specific to your situation.
Sources: Biote Corp. SEC filings and press releases (ir.biote.com); Brewer, Attorneys & Counselors press releases; SEC EDGAR (case no. biote/BTMD); FDA public statements on adverse event reporting; Dallas County 101st District Court records; Delaware Court of Chancery filings; Business Wire; StockTitan; Law360.

Comments