If you’re searching for a Liletta IUD class action settlement to file a claim with — you need to know upfront: as of 2026, no class action settlement exists for Liletta. What does exist is a growing body of individual lawsuits filed by women who suffered serious injuries, and a clear legal path to pursue your own claim if Liletta harmed you.
This guide covers everything honestly: what Liletta is, what the real risks are (straight from FDA labeling), the types of injuries women have sued over, how product liability and medical malpractice claims work, and exactly what steps to take if you believe you were hurt.
Quick Answer: Liletta (levonorgestrel 52mg) is a hormonal IUD manufactured by AbbVie and Medicines360, FDA-approved in 2015. Serious reported complications include uterine perforation, ectopic pregnancy, sepsis, and pelvic inflammatory disease leading to infertility. Women have filed individual lawsuits against the manufacturer and their doctors over these injuries. There is currently no Liletta class action settlement — anyone claiming otherwise is providing false information. If you were injured, your path forward is a personal product liability or medical malpractice claim.

What Is Liletta and Who Makes It?
Liletta is a hormonal intrauterine system (IUS/IUD) that releases levonorgestrel — a progestin hormone — directly into the uterus to prevent pregnancy. It’s shaped like a small T and inserted by a healthcare provider during an office visit. Mirena Lawsuit
| Fact | Details |
|---|---|
| Generic name | Levonorgestrel-releasing intrauterine system 52 mg |
| Brand name | LILETTA® |
| Manufacturers | AbbVie and Medicines360 |
| FDA approval date | March 2, 2015 |
| Approved uses | Pregnancy prevention (up to 8 years); heavy menstrual bleeding treatment (up to 5 years) |
| Hormone released | Levonorgestrel (LNG) |
| Initial release rate | ~20 mcg/day, decreasing over time |
| Cost advantage | Marketed as a lower-cost alternative to Mirena |
| Available since in Europe | Several years before U.S. approval |
Medicines360 is a San Francisco-based nonprofit — an unusual arrangement in the pharmaceutical world — that co-developed Liletta with the aim of making hormonal IUDs more affordable and accessible to lower-income women. AbbVie handles commercialization in the U.S.
Liletta works by thickening cervical mucus (making it harder for sperm to reach an egg), thinning the uterine lining, and in some cases affecting sperm motility. Unlike the copper Paragard IUD, Liletta does not use hormones to trigger a spermicidal copper reaction — it uses synthetic progestin.
What the FDA Says About Liletta’s Risks
This is where most online content fails women: they either ignore the actual FDA label or bury the serious risks under marketing language. The FDA’s prescribing information for Liletta — sourced directly from clinical trial data — lists several serious complications that every Liletta user should know about.
Serious Complications (Per FDA Label)
The FDA’s clinical trial data confirmed that serious adverse reactions related to Liletta include ectopic pregnancies, ovarian cysts, and IUS perforation requiring laparoscopic surgery.
| Complication | What It Means | Risk Level |
|---|---|---|
| Uterine perforation | Liletta pierces through the uterine wall, potentially migrating to other organs | Serious; may require surgery |
| Ectopic pregnancy | Pregnancy occurs outside the uterus (usually fallopian tube), which can rupture and cause life-threatening internal bleeding | Medical emergency |
| Sepsis / Group A Streptococcal infection | Severe systemic infection, particularly in the days after insertion | Can be fatal |
| Pelvic Inflammatory Disease (PID) | Infection spreading to uterus, fallopian tubes, and ovaries | Can cause permanent infertility |
| Endometritis | Infection of the uterine lining | Can cause scarring and fertility problems |
| Ovarian cysts | Fluid-filled sacs on the ovaries | Usually resolve; sometimes require treatment |
| Device expulsion | Liletta partially or fully leaves the uterus without the user knowing | Loss of contraceptive protection |
Perforation: What the Label Actually Says
Liletta’s official prescribing information states that perforation can happen at any point — not just during insertion. Delayed detection of a perforated IUD can result in migration outside the uterine cavity, adhesions, peritonitis, intestinal perforations, intestinal obstruction, and abscesses. Surgery is required to remove a perforated IUD.
The risk of perforation is specifically noted to be higher in breastfeeding women and women whose uteruses are fixed in certain positions.
Ectopic Pregnancy: The Specific Numbers
In Liletta’s clinical trial, the rate of ectopic pregnancy was approximately 0.12 per 100 women-years. That sounds small — but the FDA warns that if pregnancy occurs while Liletta is in place, it is more likely to be ectopic than intrauterine. An ectopic pregnancy can rupture, causing internal bleeding and death.
PID and Infertility
The FDA notes that Liletta is associated with an increased risk of PID, likely because organisms can be introduced into the uterus during insertion. In the clinical study, cases of PID and endometritis were diagnosed in participants — some within days of insertion, others more than six months later. PID can cause permanent tubal damage, leading to ectopic pregnancy or infertility.
Common Side Effects (The Non-Emergency Issues)
Not every Liletta problem rises to the level of a lawsuit. The clinical trial also tracked common side effects that caused participants to discontinue use:
| Side Effect | Discontinuation Rate in Clinical Trial |
|---|---|
| Device expulsion | 4.1% |
| Bleeding complaints | 2.5% |
| Acne | 1.4% |
| Dysmenorrhea (painful periods) | 1.0% |
| Weight increase | 1.0% |
| Mood swings | 0.8% |
| Uterine spasm | 0.7% |
| Dyspareunia (painful intercourse) | 0.6% |
| Pelvic pain | 0.6% |
Irregular bleeding and spotting are especially common in the first 3 to 6 months. Periods may then become shorter or lighter — or stop altogether.
Depression and mood changes are also reported by some users, though Liletta’s official labeling does not carry the same prominent depression warning that Mirena’s label includes.
Why People Are Filing Lawsuits Against Liletta
Individual lawsuits against Liletta’s manufacturer and against treating physicians have been filed across the country. These are not class actions — they are individual personal injury cases, each evaluated on its own facts. Here’s what the allegations typically involve:
Product Liability Claims Against the Manufacturer
A product liability lawsuit argues that the device itself was defective, or that the manufacturer didn’t adequately warn patients and doctors about risks. For Liletta, the legal theories most commonly pursued include:
Failure to Warn. Even though Liletta’s website is considered more forthcoming than some other IUD manufacturers about perforation risk, plaintiffs have argued that warnings are buried in fine print and are not presented in a way that gives women a genuinely informed choice. Liletta’s site acknowledges that the device can “go through” the uterine wall — but critics argue these disclosures are still insufficient given the severity of the outcomes.
Design Defect. If a plaintiff can show that the device’s design made perforation, embedment, or other injuries more likely than a reasonably designed alternative, this can support a design defect claim.
Breast Cancer Allegations. Some individual lawsuits have alleged that Liletta’s levonorgestrel increases breast cancer risk. A large Danish study published in JAMA found that women using levonorgestrel IUDs had roughly a 40% higher chance of a breast cancer diagnosis compared to non-users — though the absolute increase was small (about 14 additional cases per 10,000 women). Proving causation in these cases has been legally difficult, because courts require expert testimony establishing that the IUD — and not other risk factors — caused a particular plaintiff’s cancer.
Medical Malpractice Claims Against Doctors
Not every Liletta injury is the manufacturer’s fault. In some cases, the doctor’s conduct during insertion or removal may be the main issue. Medical malpractice claims may be appropriate when:
- A provider inserted Liletta in a woman with contraindications (like active PID or certain uterine abnormalities)
- Insertion technique was improper, increasing perforation risk
- The provider failed to diagnose a perforation or expulsion in a timely way, leading to additional injury
- A provider dismissed a patient’s complaints of pain or infection, delaying treatment
A medical malpractice case requires showing that the provider’s care fell below the accepted standard — typically through expert medical testimony from another physician.
How Liletta Lawsuits Differ from the Paragard MDL
Many women searching online confuse Liletta lawsuits with the massive Paragard IUD multidistrict litigation (MDL). It’s important to understand the difference:
| Feature | Liletta Lawsuits | Paragard MDL |
|---|---|---|
| Device type | Hormonal (levonorgestrel) | Non-hormonal (copper) |
| Manufacturer | AbbVie / Medicines360 | CooperSurgical / Teva Pharmaceuticals |
| Legal structure | Individual lawsuits | MDL No. 2974 (3,800+ cases, N.D. Georgia) |
| Core allegation | Perforation, ectopic pregnancy, PID, breast cancer risk | Device breakage during removal |
| Settlement status | No settlement | No global settlement; bellwether trials began 2026 |
| Class action settlement | None exists | None exists (litigation ongoing) |
The Paragard litigation involves a specific, well-documented design failure — the device breaking during routine removal, leaving fragments inside patients. Liletta lawsuits involve different mechanisms of injury and are currently litigated individually rather than consolidated into a federal MDL. Power Home Remodeling Group Class Action Lawsuit
This matters for you because Paragard plaintiffs are part of a coordinated federal proceeding with shared legal resources. If you were injured by Liletta, you would be filing a standalone lawsuit — which means choosing an attorney experienced in individual product liability cases, not simply joining an existing MDL.
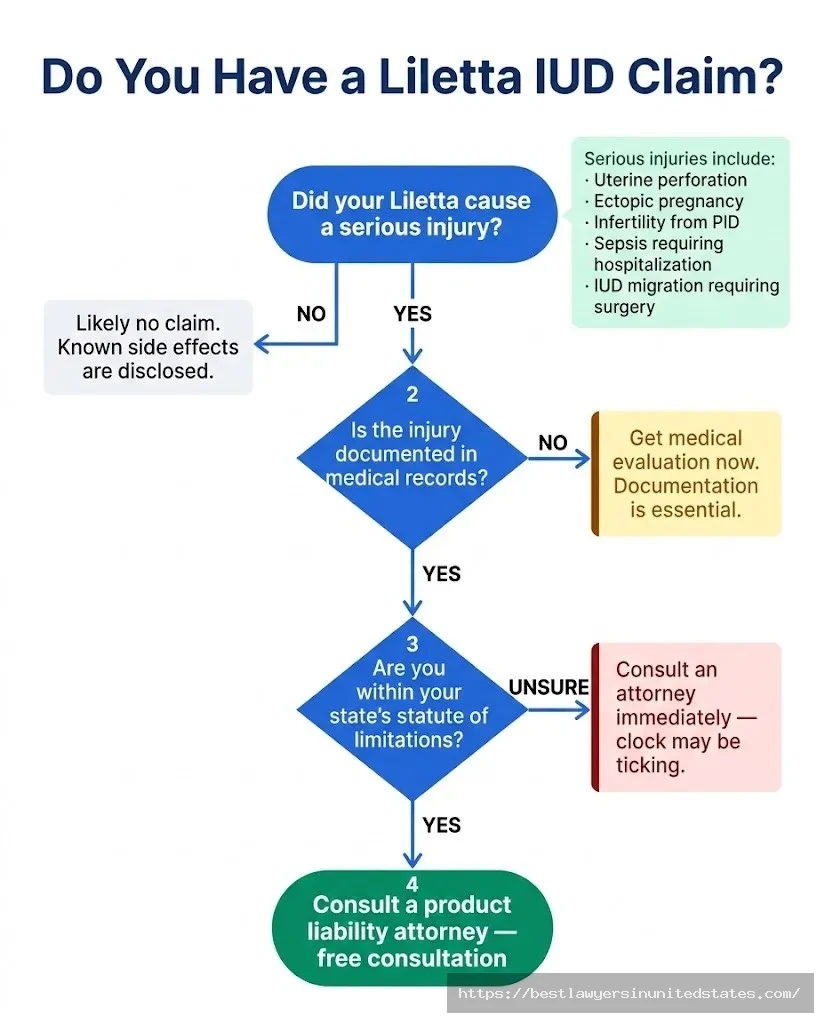
Do You Have a Liletta Claim? Key Questions to Ask
Before speaking with an attorney, work through these questions honestly:

Is Your Injury Listed in the Warnings?
This is the critical threshold question. If your injury is a known, disclosed risk — irregular bleeding, minor pelvic discomfort, mood changes — you likely don’t have a viable product liability claim, because the manufacturer warned about those outcomes. Courts generally don’t hold manufacturers liable for harms they clearly disclosed.
However, just because something is mentioned in a label doesn’t automatically defeat your claim. If the warning was so vague or buried that a reasonable person couldn’t understand the actual risk, or if the injury happened in circumstances the label didn’t address, there may still be grounds to sue.
Did You Suffer a Serious, Documentable Harm?
Lawsuits are expensive to pursue, and attorneys who take these cases on contingency (no fee unless you win) need to see significant damages. Injuries that typically support viable claims include:
- Uterine perforation requiring surgery (laparoscopy or open surgery)
- Ectopic pregnancy requiring surgery, especially with loss of the fallopian tube
- PID or endometritis resulting in documented infertility
- Sepsis requiring hospitalization
- IUD migration requiring complex surgical removal
- Breast cancer diagnosis in someone with low other risk factors
Minor or temporary side effects that resolved without serious medical intervention are unlikely to support a lawsuit worth pursuing.
Was There Medical Documentation?
Your medical records are everything in a product liability or malpractice case. The more clearly your medical records document what happened, when it happened, how it was treated, and what the long-term effects are — the stronger your claim. Gaps in documentation don’t kill a case, but they create challenges.
Are You Within the Statute of Limitations?
Every state has a statute of limitations for personal injury claims — the deadline after which you can no longer sue. These vary by state, typically running from 2 to 4 years from the date of injury (or from when you knew or should have known that the device caused your injury). If you were injured years ago and haven’t acted, speak with an attorney immediately — time may be running out.
What Compensation Can You Pursue?
If your claim succeeds, you may be able to recover damages for:
| Damage Type | What It Covers |
|---|---|
| Medical expenses | Hospital bills, surgery costs, follow-up treatment, ongoing care |
| Future medical costs | Additional procedures, fertility treatments, long-term care needs |
| Lost wages | Income missed due to recovery, hospitalization, ongoing disability |
| Loss of future earning capacity | If injury affects your ability to work long-term |
| Pain and suffering | Physical pain and emotional distress caused by the injury |
| Loss of fertility | Significant damages when injury results in permanent infertility |
| Loss of consortium | Impact on spousal relationship (in some states) |
| Punitive damages | In rare cases where manufacturer conduct was especially egregious |
Settlement amounts in individual IUD injury cases vary enormously based on the severity of the injury, the strength of the evidence, the state where the case is filed, and negotiation. There is no published average or “standard” payout for Liletta claims — each case is unique.
How to Pursue a Liletta Claim: Step-by-Step
Step 1: Get Medical Attention First
If you’re still experiencing symptoms — pain, irregular bleeding, signs of infection, or concerns about your IUD’s position — see a doctor before doing anything else. Your health comes first. Also, from a legal standpoint, prompt medical treatment creates contemporaneous documentation that connects your symptoms to the device.
Step 2: Gather and Preserve Your Medical Records
You’ll want everything you can find:
- Records from the clinic or practice where Liletta was inserted (including the lot number of the device if possible)
- Any ultrasound records showing device position
- Emergency room or hospital records related to complications
- Records from any surgeries you had
- Records from your OB/GYN, primary care provider, or specialist relating to your symptoms
- Documentation of any infertility treatment you’ve needed
You have a legal right to your own medical records. Request them in writing from each provider.
Step 3: Preserve the Device (If Possible)
If Liletta was removed, ask the provider to preserve it. In some cases, the physical device can be tested or examined as evidence. This isn’t always possible — especially if it was removed during an emergency — but ask.
Step 4: Document Your Damages
Keep a running record of:
- Every medical bill and expense
- Missed work and lost income
- A journal of how your injury has affected your daily life, physical health, and emotional wellbeing
- Any out-of-pocket costs for treatment
Step 5: Consult a Product Liability or Medical Malpractice Attorney
This is not optional if you want to pursue a claim. Liletta lawsuits require expert medical testimony and substantial legal resources. Attorneys who handle these cases work on contingency — meaning you pay nothing unless they win. Look for attorneys with specific experience in:
- Pharmaceutical or medical device product liability
- Women’s health injury cases
- IUD-related litigation
Initial consultations are typically free. Be prepared to share your timeline, your medical records summary, and a description of your injuries. The attorney will tell you honestly whether your case has merit. Aqua Finance Lawsuit
Step 6: Act Before the Statute of Limitations Expires
Once you know you were injured, the clock is ticking. Don’t wait. Even if you’re not sure whether you have a case, a free consultation with a product liability attorney will clarify whether you need to act now. Statutes of limitations in personal injury cases are hard deadlines — courts don’t extend them simply because you didn’t know about the law.
Should You Report Your Injury to the FDA?
Yes — and this is something many injured women don’t know they can do. You can report Liletta side effects and complications directly to the FDA through MedWatch, the FDA’s safety reporting system, at fda.gov/medwatch or by calling 1-800-FDA-1088.
Reporting doesn’t create a lawsuit and doesn’t require an attorney. But FDA adverse event data helps regulators identify patterns that individual women can’t see — if enough reports come in showing a particular type of injury, the FDA may require a label change or issue a safety communication. Your report could protect future patients.
Frequently Asked Questions
Is there a Liletta IUD class action settlement I can file a claim with?
No. As of March 2026, there is no Liletta class action settlement, no claim deadline, and no settlement administrator distributing payments. Any website claiming otherwise is providing false information to drive legal lead generation. Individual lawsuits against Liletta’s manufacturer have been filed, but no class action has been certified and no settlement has been reached.
Who manufactures Liletta?
Liletta is manufactured by AbbVie and Medicines360. Medicines360 is a San Francisco-based nonprofit that co-developed the device. AbbVie handles U.S. commercialization. The trademark is registered to Odyssea Pharma SPRL, an AbbVie company.
What are the most serious complications reported with Liletta?
Per the FDA’s prescribing information and clinical trial data, the most serious complications are uterine perforation (which may require laparoscopic surgery), ectopic pregnancy (a medical emergency that can be life-threatening), sepsis (including Group A streptococcal sepsis), and pelvic inflammatory disease — which can cause permanent infertility.
Can I sue for Liletta side effects like acne, mood changes, or irregular bleeding?
These are disclosed, known side effects listed in Liletta’s label. Courts generally don’t hold manufacturers liable for harms that were clearly warned about. To have a viable lawsuit, you typically need a serious injury — like perforation, ectopic pregnancy, or infertility — that caused significant, documentable harm.
How is a Liletta lawsuit different from the Paragard MDL?
The Paragard MDL involves thousands of cases consolidated in federal court in Georgia, all concerning the same issue: the copper Paragard device breaking during removal. There is no Liletta MDL. Liletta lawsuits are filed individually, in state or federal court, by each injured plaintiff.
Do I need a lawyer to sue?
To pursue a product liability or medical malpractice case against a pharmaceutical manufacturer, you practically need a lawyer. These cases require expert testimony, extensive discovery, and resources that no individual can realistically manage alone. Fortunately, product liability attorneys handle these cases on contingency, so you pay nothing upfront.
What if my injury happened years ago?
It depends on your state’s statute of limitations. Some states count the clock from the date of injury; others from the date you discovered (or reasonably should have discovered) that the IUD caused your harm. Some states also have statutes of repose that create hard outer limits. If you were injured more than two years ago, consult an attorney immediately — time may already be running out.
Can breastfeeding women use Liletta?
Liletta is not absolutely contraindicated during breastfeeding, but the FDA specifically notes that the risk of perforation is increased in breastfeeding women. Insertion should typically be delayed until at least 4 weeks postpartum, or longer if the uterus has not fully returned to its normal position.
What if my doctor dismissed my pain or symptoms?
If a provider failed to respond appropriately to your complaints and delayed diagnosis of a serious complication — increasing your harm — that conduct may support a medical malpractice claim separate from (or in addition to) any product liability claim against the manufacturer. Discuss the full timeline of your care with an attorney.
Is there a link between Liletta and breast cancer?
Research exists suggesting that levonorgestrel IUDs (including Liletta, Mirena, Kyleena, and Skyla) may modestly increase breast cancer risk. A large JAMA-published study found roughly a 40% increased relative risk, though the absolute increase was small. Some individual lawsuits have been filed on this basis, but proving causation in any individual case is scientifically and legally difficult. This is an evolving area and one worth discussing with both your doctor and an attorney if you have been diagnosed with breast cancer after using Liletta.
How do I report a Liletta injury to the FDA?
Visit fda.gov/medwatch or call 1-800-332-1088. You can also ask your healthcare provider to submit a report. Reporting is voluntary, free, and does not require legal representation.
The Bottom Line
Liletta is a widely used hormonal IUD that works as intended for most women. But for some, it has caused serious, life-altering injuries — uterine perforation, ectopic pregnancy, infertility-inducing infections, and more. Those injuries are real, and the legal system provides a path to seek accountability and compensation.
What doesn’t exist — despite what many search results imply — is a ready-made class action settlement waiting for you to file a claim. The honest reality is that Liletta injury claims are individual matters, evaluated case by case, pursued through product liability and medical malpractice law.
If you were hurt by Liletta, the steps are clear: document your injury, gather your records, and speak with a product liability attorney who handles pharmaceutical device cases. Most offer free consultations and take cases on contingency. You have nothing to lose by making that call — and potentially significant compensation to gain if your case has merit.
