The Mirena IUD lawsuit saga is one of the most complex medical device legal battles in recent U.S. history. Bayer Healthcare’s hormone-releasing intrauterine device has been the subject of tens of thousands of FDA injury reports and thousands of lawsuits alleging that Bayer failed to warn women about serious risks — including organ perforation, dangerous brain pressure, and a potential link to breast cancer. Bayer settled approximately 4,000 perforation lawsuits for $12.2 million in 2018, but the litigation has evolved significantly since then, with new claims emerging as recently as 2024. If you used Mirena and suffered serious side effects, this guide covers everything you need to know about what happened, what was settled, and what legal options may still exist. Power Home Remodeling Group Class Action Lawsuit
Quick Answer: The original Mirena MDL (multidistrict litigation) lawsuits over organ perforation and brain pressure conditions have largely concluded. Bayer paid $12.2 million to settle roughly 4,000–4,800 perforation claims in 2018. The pseudotumor cerebri (brain pressure) MDL was dismissed after judges rejected plaintiffs’ expert witnesses. A separate breast cancer class action was filed in 2022 but was voluntarily dismissed in May 2024. As of early 2026, most attorneys have stopped accepting perforation/migration cases. However, new individual lawsuits may still be possible for women with recent diagnoses — particularly involving breast cancer links. Read on for the full picture.

Mirena Lawsuit Overview
| Key Fact | Details |
|---|---|
| Product | Mirena IUD (levonorgestrel-releasing intrauterine system) |
| Manufacturer | Bayer Healthcare Pharmaceuticals |
| FDA Approval | 2000 |
| Injury Reports Filed with FDA | 70,000+ |
| Primary MDL Location | U.S. District Court, Southern District of New York |
| Secondary Litigation | Bergen County, New Jersey (MCL No. 297) |
| 2018 Perforation Settlement | $12.2 million (approx. 4,000–4,800 plaintiffs) |
| MDL Status (Perforation) | Closed December 8, 2020 |
| MDL Status (Pseudotumor Cerebri) | Dismissed |
| NJ MCL Status | Closed April 2021 |
| Breast Cancer Class Action | Filed March 2022; dismissed May 2024 |
| Current Attorney Acceptance | Most firms no longer accepting cases |
What Is the Mirena Lawsuit About?
Background of the Mirena IUD
Mirena is a small, T-shaped hormonal intrauterine device manufactured by Bayer Healthcare Pharmaceuticals. It was first introduced in Finland in 1990 and approved by the U.S. Food and Drug Administration in 2000. The device works by releasing a steady low dose of levonorgestrel — a synthetic progestin hormone — directly into the uterus. It’s approved both as a long-term contraceptive (lasting up to 8 years with newer labeling) and as a treatment for heavy menstrual bleeding.
By Bayer’s own estimates, Mirena became the most-prescribed IUD in the United States. Millions of women were implanted with the device after its U.S. launch. The FDA has since received more than 70,000 adverse event reports tied to Mirena, making it one of the most complaint-heavy medical devices in modern regulatory history.
Problems began surfacing not long after Mirena hit the U.S. market. Women reported that the device was moving inside their bodies — sometimes migrating through the uterine wall and into the abdominal cavity, puncturing organs including the bladder, bowel, fallopian tubes, and ovaries. Others reported a devastating neurological condition called pseudotumor cerebri (also called idiopathic intracranial hypertension), which causes dangerous pressure to build up around the brain. Later, research raised red flags about a potential link between long-term Mirena use and breast cancer — a risk Bayer did not disclose on its original labeling.
The core of every Mirena lawsuit was the same allegation: Bayer knew these risks existed and chose not to tell women or their doctors.
Timeline of Key Events in Mirena Litigation
| Date | Event | Details |
|---|---|---|
| 2000 | FDA approval | Mirena cleared for contraceptive use in the U.S. |
| 2009 | FDA warning letter | FDA accused Bayer of overstating benefits and understating risks in marketing materials |
| 2013 | First MDL formed | JPML consolidated perforation/migration lawsuits into MDL 2434, S.D.N.Y. |
| 2013 | NJ MCL formed | NJ Supreme Court consolidated similar cases into MCL No. 297, Bergen County |
| 2014 | Finnish study published | Obstetrics & Gynecology study found elevated breast cancer risk in Mirena users |
| 2017 | Pseudotumor cerebri MDL | Second MDL formed in S.D.N.Y. for intracranial hypertension claims |
| April 2018 | Perforation settlement | Bayer agreed to settle ~4,000–4,800 cases for $12.2 million |
| 2018–2020 | Expert witness battles | Judges excluded plaintiffs’ experts in pseudotumor cerebri cases |
| December 8, 2020 | Final MDL closed | Both New York MDLs formally closed |
| April 2021 | NJ MCL closed | New Jersey multicounty litigation concluded |
| March 14, 2022 | Breast cancer class action filed | Priya Sidhu filed against Bayer in N.D. California (Case No. 5:22-cv-01603) |
| October 2023 | Class action survives dismissal | Federal judge rejected Bayer’s motion to dismiss |
| May 2024 | Breast cancer case dismissed | Parties filed stipulation of voluntary dismissal |
| October 2024 | New breast cancer study | Danish Cancer Institute found 40% higher breast cancer risk in levonorgestrel IUD users |
| December 2025 | Status update | Most law firms no longer accepting Mirena cases |
Who Filed the Lawsuits?
The earliest Mirena lawsuits were brought by individual plaintiffs across the country who experienced organ perforation and IUD migration. When the volume of similar cases grew large enough, the federal Judicial Panel on Multidistrict Litigation (JPML) consolidated them into two MDLs in the Southern District of New York. Law firms representing plaintiffs included some of the country’s leading mass tort litigation practices.
The most recent major legal action was filed by Priya Sidhu, represented by the firm Bursor & Fisher, P.A. Her 2022 complaint named Bayer U.S. LLC as the defendant and sought class action status on behalf of all U.S. women implanted with Mirena, centered on allegations that Bayer concealed known breast cancer risks.
The named defendant in all Mirena litigation is Bayer Healthcare Pharmaceuticals, Inc., the U.S. subsidiary of the German pharmaceutical giant Bayer AG.
What Are the Core Allegations?
Every strand of Mirena litigation shared a common thread: Bayer put profits ahead of patient safety.
Key allegations across Mirena lawsuits include:
- ✅ Failure to warn — Bayer did not adequately disclose the risk that Mirena could migrate from its implanted position and perforate the uterus or other organs
- ✅ Defective design — Plaintiffs alleged the device was inherently prone to migration after insertion, not just during the procedure
- ✅ Concealment of breast cancer risk — Studies dating back to at least 2010 showed elevated breast cancer risk; Bayer allegedly knew and did not update its labeling
- ✅ Misleading marketing — In 2009, the FDA sent Bayer a formal warning letter for promotional events that promised Mirena would improve relationships and sex lives, while downplaying side effects including irregular bleeding, ovarian cysts, and weight gain
- ✅ Failure to disclose pseudotumor cerebri risk — Women experiencing dangerous intracranial pressure said Bayer’s pre-2014 label had no meaningful warning about this risk
- ✅ Deceptive consumer practices — The California breast cancer class action alleged violations of the state’s Unfair Competition Law and consumer protection statutes
Who Qualified — and Who May Still Qualify — for Mirena Compensation?
Quick Answer: Past Mirena settlements have already closed. If you suffered organ perforation or migration, the MDL settlement pool is no longer available. However, if you have a new diagnosis connected to Mirena use — particularly breast cancer — and you are within your state’s statute of limitations, you may still be able to pursue an individual lawsuit. Contact an attorney immediately to assess your options.
Eligibility for the 2018 Perforation Settlement (Now Closed)
The 2018 Bayer settlement covered women who experienced organ perforation or IUD migration that caused documented physical injuries. That settlement pool is no longer accepting claims. The cases were fully resolved and the MDLs closed.
Who May Still Have Legal Options in 2026
| Potential Claim Type | Status | Notes |
|---|---|---|
| Organ perforation / uterine migration | MDL closed; very limited options | Few attorneys accepting these cases |
| Pseudotumor cerebri / intracranial hypertension | MDL dismissed | Extremely difficult to pursue |
| Breast cancer diagnosis linked to Mirena use | Active area of inquiry | New research strengthens potential claims; consult attorney |
| Fraudulent concealment of cancer risk | Being evaluated | Statute of limitations may be extended if you only recently learned of the risk |
Who Does NOT Have Current Claim Options
❌ Women who already received compensation from the 2018 perforation settlement and signed a release ❌ Women whose perforation cases were dismissed on the merits or for statute of limitations ❌ Anyone who opted out of the MDL without filing an individual claim before applicable deadlines ❌ Women whose cases were part of the pseudotumor cerebri MDL that was dismissed (barring successful appeal pathways)
The Breast Cancer Question — A Growing Legal Frontier
This is where new legal activity is most likely to emerge. In 2014, a Finnish study published in Obstetrics & Gynecology found that women using Mirena to treat heavy periods faced an elevated breast cancer risk. In October 2024, researchers from The Danish Cancer Institute published a study of nearly 80,000 women between the ages of 15 and 49 that found levonorgestrel IUD users had a 40% higher risk of breast cancer compared to non-users.
The 2022 class action alleged Bayer knew about a 20–30% elevated risk and deliberately left it off the label. Although that particular class action was voluntarily dismissed in May 2024, the new Danish research is prompting attorneys to re-evaluate individual breast cancer claims. If you used Mirena and were later diagnosed with breast cancer — especially hormone receptor-positive (ER/PR positive) cancer — this may be legally significant.
Required Documentation to Explore a Claim
| Document | Why It Matters | Where to Find It |
|---|---|---|
| Medical records of Mirena insertion | Proves you used the device and when | Your OB-GYN or women’s health provider |
| Medical records of injury or diagnosis | Documents what went wrong | Your treating physician or hospital records |
| Imaging / surgical reports | Confirms perforation, migration, or complications | Radiologist and surgeon records |
| Pharmacy/insurance records | Can substitute if original records are incomplete | Health insurer, pharmacy |
| Cancer diagnosis records | Pathology reports, oncology notes | Oncologist, hospital pathology department |
What Did the Mirena Settlement Pay Out?
Quick Answer: Bayer settled approximately 4,000–4,800 perforation lawsuits for a total of $12.2 million in 2018. Individual payouts were confidential and varied based on injury severity. The pseudotumor cerebri cases were dismissed without any Bayer payout. There is no currently open settlement fund.
The 2018 Perforation Settlement Breakdown
| Category | Details |
|---|---|
| Total Settlement Amount | $12.2 million |
| Estimated Cases Covered | ~4,000–4,800 plaintiffs |
| Cases Included | MDL 2434 (S.D.N.Y.), NJ MCL No. 297, Missouri, California cases |
| Cases Excluded | Pseudotumor cerebri claims, ectopic pregnancy claims |
| Individual Amounts | Confidential (varied by injury severity) |
| Payment Form | Confidential; standard mass tort settlement structure |
Why Individual Payouts Varied
In a mass tort settlement like this one, no two payouts are identical. Here’s what drove the differences in Mirena perforation settlement amounts:
Factors that increased settlement value:
- Severity of the organ perforation and number of organs affected
- Surgical interventions required for removal (laparoscopy, open surgery)
- Long-term health consequences (infertility, chronic pain, bowel injury)
- Lost wages and lost earning capacity
- Pain and suffering documentation
- State of residency (some states have more plaintiff-friendly tort laws)
Factors that decreased settlement value:
- Minimal documented injuries
- Delayed diagnosis (gap between injury and treatment)
- Pre-existing conditions that complicated causation
Pseudotumor Cerebri Cases — No Settlement Reached
The second major group of lawsuits — involving women who developed pseudotumor cerebri (also called idiopathic intracranial hypertension or IIH) — ended without any settlement. Judges in the Southern District of New York ruled that the expert witnesses presented by plaintiffs did not meet the Daubert standard for admissible scientific testimony. Without credible expert testimony linking Mirena to their brain pressure condition, plaintiffs could not prove causation, and the cases were dismissed.
This doesn’t mean these women weren’t harmed. It means the evidence standard required in federal court wasn’t met with the science available at the time.
Current Status: Where the Mirena Lawsuit Stands in 2026
The Perforation and Migration MDL — Closed
The final Mirena MDL closed formally on December 8, 2020. The New Jersey multicounty litigation wrapped up by April 2021. These cases are over. If you were injured by Mirena perforation or migration and didn’t file before those proceedings closed, your window to participate in group litigation has passed. Whether you have any remaining options depends entirely on your state’s statute of limitations and specific circumstances — which is why talking to an attorney quickly matters.
The Breast Cancer Class Action — Voluntarily Dismissed, But Not Dead
The 2022 breast cancer class action (Sidhu v. Bayer U.S. LLC, Case No. 5:22-cv-01603) was a significant development. It survived Bayer’s motion to dismiss in October 2023, which meant a federal judge agreed the claims were legally plausible enough to proceed. But in May 2024, the parties filed a stipulation of voluntary dismissal — ending that particular case.
The voluntary dismissal doesn’t necessarily mean the breast cancer theory is dead. It may reflect strategic litigation decisions, ongoing settlement discussions, or a plan to refile differently. The October 2024 Danish study, which found a 40% elevated breast cancer risk, gives potential plaintiffs new ammunition that wasn’t available in 2022.
What Attorneys Are Saying Right Now
As of December 2025, most mass tort law firms have stopped accepting Mirena cases — particularly for perforation and migration. The litigation closed with results that weren’t favorable to plaintiffs. However, a smaller number of attorneys are still evaluating individual claims, especially for:
- Women recently diagnosed with breast cancer who were long-term Mirena users
- Women with documented complications that occurred more recently (within applicable statute of limitations)
- Women who can argue their claim was “tolled” (paused) because they only recently discovered the connection between Mirena and their injury
Recent Developments (2024–2026)
- October 2024: Danish Cancer Institute published a landmark study of ~80,000 women showing a 40% elevated breast cancer risk in levonorgestrel IUD users — the most comprehensive research yet linking the hormone in Mirena to cancer risk
- May 2024: Sidhu breast cancer class action voluntarily dismissed after surviving Bayer’s motion to dismiss — leaving the legal question open for individual claims
- December 2025: Drugwatch and other litigation monitoring sites confirm most attorneys have ceased accepting new Mirena MDL cases
- 2026: Legal observers are watching whether new individual breast cancer suits emerge and whether attorneys will attempt a new consolidated action based on the stronger 2024 Danish research
How to File a Mirena Lawsuit — If You Still Have Options
Because there is no currently open class action claim portal, the path to pursuing a Mirena-related claim in 2026 is through an individual product liability lawsuit, not a mass claim form. This is a more involved process but can result in higher individual compensation if successful.

Step 1: Assess Whether You’re Within the Statute of Limitations
This is the most time-sensitive step. Each state has its own deadline for filing personal injury or product liability claims — typically 2 to 4 years. The clock often starts when you knew or should have known that the device caused your injury. If you only recently learned about the breast cancer link, a “discovery rule” may give you additional time. Aqua Finance Lawsuit: Complete 2026 Guide
Don’t assume you’ve missed the deadline without consulting an attorney first.
Step 2: Gather Your Medical Records
Start collecting documentation now, even before you speak to a lawyer. You’ll want:
- Mirena insertion records (date, provider, model/lot number if available)
- Records of any complications, ER visits, or imaging
- Surgical records if the device was removed surgically
- Oncology records and pathology reports if you were diagnosed with cancer
- Health insurance records and Explanation of Benefits (EOBs)
Step 3: Find an Attorney Who Handles Medical Device Cases
Look for a mass tort or product liability attorney with experience in medical device litigation specifically. Many offer free initial consultations. At that consultation, they will review your records and tell you whether you have a viable claim worth pursuing.
Questions to ask any attorney:
- Have you handled Bayer/Mirena cases or similar IUD device cases?
- What is your assessment of my statute of limitations?
- Do you work on contingency (meaning no fees unless you win)?
- What are the realistic outcomes for my type of claim?
Step 4: File Your Complaint
If the attorney takes your case, they will prepare and file a formal legal complaint. This is different from submitting a claim form in a class action. Filing a complaint initiates a lawsuit.
Step 5: Discovery and Expert Testimony
Your attorney will build your case by obtaining Bayer’s internal documents, commissioning expert medical testimony linking your injury to Mirena, and deposing Bayer representatives. The strength of expert testimony is what made or broke the prior Mirena MDLs — so finding qualified experts is critical.
Step 6: Settlement Negotiation or Trial
Most product liability cases settle before trial. Your attorney will negotiate with Bayer’s legal team. If no fair settlement is reached, your case goes to trial. The entire process can take 1–4 years depending on complexity.
Critical Deadlines to Know
| Deadline Type | What It Means | Action Required |
|---|---|---|
| Statute of Limitations | Last day to file your lawsuit | Varies by state — typically 2–4 years from discovery of injury |
| Statute of Repose | Hard cutoff in some states regardless of discovery | Varies — some states have 10-year hard limits |
| Evidence Preservation | Time-sensitive — records can be lost | Begin gathering records NOW |
| Attorney Consultation | The sooner the better | Contact an attorney immediately if you think you have a claim |
Mirena Lawsuit vs. Similar IUD Litigation
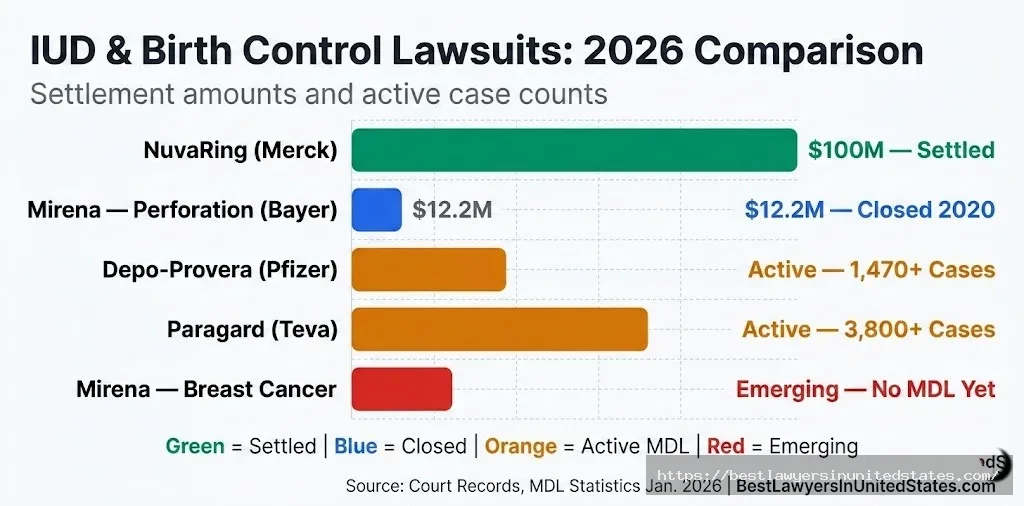
How Mirena Cases Compare to Current and Recent IUD Lawsuits
| Lawsuit | Device | Manufacturer | Settlement / Status | Primary Injury |
|---|---|---|---|---|
| Mirena Lawsuit | Hormonal IUD | Bayer | $12.2M settled (2018); MDLs closed | Perforation, pseudotumor cerebri, breast cancer |
| Paragard IUD Lawsuit | Copper IUD | Teva / CooperSurgical | Active MDL; ~3,800 cases as of Jan. 2026 | Device breakage during removal |
| NuvaRing Lawsuit | Hormonal ring | Merck | $100M settlement | Cardiovascular events, blood clots |
| Essure Lawsuit | Permanent contraceptive | Bayer | Settled; product recalled 2018 | Organ perforation, migration, pain |
| Depo-Provera Lawsuit | Injectable birth control | Pfizer | Active MDL; 1,470+ cases | Meningioma (brain tumor) |
What Makes the Mirena Litigation Distinctive
The Mirena lawsuits are notable for a few reasons. First, the volume of FDA adverse event reports — over 70,000 — was exceptionally high for a single device. Second, the litigation played out in a way that’s uncommon in mass tort cases: the MDLs ultimately favored Bayer because of the expert witness issue, not because the injuries weren’t real. Third, the emerging breast cancer angle introduces a new theory of harm that wasn’t the focus of the original litigation, giving some women who might have missed the first wave of lawsuits a potential new avenue.
The Paragard and Depo-Provera litigations are currently more active and represent where mass tort IUD/birth control legal activity is most concentrated in 2026.
Do You Need a Lawyer for a Mirena Claim?
Quick Answer: For Mirena in 2026, yes, you need an attorney. Unlike a standard class action settlement with a public claim form, pursuing a Mirena claim today requires filing an individual product liability lawsuit. This is complex litigation that requires legal expertise, especially given the history of expert witness battles in this space.
Why Self-Filing Doesn’t Work Here
Class action claims are designed for self-filing — you fill out a form, submit documentation, and wait for a check. Mirena’s open settlement phase is over. Individual lawsuits involve formal court filings, discovery processes, depositions, and expert medical testimony. Attempting this without legal representation would be extremely difficult and likely unsuccessful.
What a Mass Tort Attorney Does for You
A qualified product liability attorney will handle every step: assessing whether your claim is timely, gathering evidence, finding expert witnesses (the critical factor in Mirena litigation), negotiating with Bayer, and representing you at trial if needed. Most work on contingency — meaning they only get paid if you win or settle, typically 30–40% of the recovery.
When to Contact an Attorney Right Now
Talk to an attorney immediately if any of the following apply:
- You used Mirena for an extended period and were later diagnosed with breast cancer (especially ER/PR positive hormone receptor-positive cancer)
- You experienced organ perforation or migration within the last 2–4 years and haven’t filed a claim
- You developed severe, chronic intracranial pressure while using Mirena
- You only recently discovered a possible connection between your health condition and Mirena
For attorney referrals, you can contact: admin@bestlawyersinunitedstates.com
Frequently Asked Questions
What is the Mirena lawsuit?
Quick Answer: The Mirena lawsuit is litigation against Bayer Healthcare Pharmaceuticals alleging that its Mirena IUD caused serious injuries — including organ perforation, dangerous brain pressure, and possibly breast cancer — and that Bayer failed to warn women or their doctors about these risks.
The lawsuits span more than a decade. The largest wave involved women whose Mirena IUD migrated from its original position and perforated the uterus or other organs. A second group involved women who developed pseudotumor cerebri, a condition causing dangerous intracranial pressure. Most recently, lawsuits have focused on an alleged link between Mirena and breast cancer risk.
Is there a current Mirena class action settlement I can file for?
Quick Answer: No. As of early 2026, there is no open Mirena class action settlement with a public claim form. The perforation MDLs closed in 2020–2021, and the breast cancer class action was voluntarily dismissed in May 2024.
If you think you have a Mirena-related injury, your path forward is an individual lawsuit, not a class action claim form. An attorney can evaluate whether you have a viable case.
How much did Bayer pay in Mirena settlements?
Quick Answer: Bayer paid approximately $12.2 million to settle around 4,000–4,800 organ perforation and migration cases in 2018. Individual payouts were confidential and varied significantly based on injury severity.
The pseudotumor cerebri cases received no Bayer payment — those cases were dismissed after judges excluded plaintiffs’ expert witnesses. There is no public record of a global, all-encompassing Mirena settlement figure.
What injuries qualify for a Mirena lawsuit?
Quick Answer: Past MDL claims focused on organ perforation, IUD migration, and pseudotumor cerebri. Going forward, breast cancer diagnoses in long-term Mirena users are the most active area of legal inquiry.
Other documented injuries that may support a claim include: ectopic pregnancy resulting from IUD failure, infertility caused by device migration, chronic pain from surgical removal, severe intracranial hypertension, and vision loss caused by optic nerve pressure.
Is there a link between Mirena and breast cancer?
Quick Answer: Research suggests yes. Multiple studies, including a 2024 Danish Cancer Institute study of nearly 80,000 women, found a 40% elevated breast cancer risk in users of levonorgestrel IUDs like Mirena compared to non-users.
The 2022 class action lawsuit alleged Bayer was aware of a 20–30% elevated risk based on earlier studies going back to at least 2010 and chose not to include that information on Mirena’s labeling. While that class action was dismissed in 2024, the newer research is stronger, and individual lawsuits based on breast cancer diagnoses may still be viable.
Why were so many Mirena lawsuits dismissed?
Quick Answer: The pseudotumor cerebri cases were dismissed because the judges overseeing them applied the Daubert standard and rejected the expert witnesses plaintiffs offered to prove causation.
In federal mass tort litigation, expert witness testimony is everything. If a judge decides that a plaintiff’s medical expert doesn’t meet the scientific rigor required by Daubert v. Merrell Dow Pharmaceuticals, that expert can’t testify — and without expert testimony establishing that Mirena caused the injury, plaintiffs can’t prove their case. This is what happened in the Mirena brain pressure MDL, and it’s what makes the breast cancer cases so important: researchers now have much more robust epidemiological data to support claims than existed in 2013.
Can I still file a Mirena lawsuit in 2026?
Quick Answer: It depends on your state’s statute of limitations and the nature of your injury. If you experienced organ perforation years ago and never filed, it’s likely too late for that claim. If you have a recent breast cancer diagnosis you believe is linked to Mirena, you may still be within your filing window.
Call a mass tort attorney for a free consultation — they can run a statute of limitations analysis specific to your state and circumstances. Don’t assume your window has closed without checking.
What is pseudotumor cerebri and how does it relate to Mirena?
Quick Answer: Pseudotumor cerebri (also called idiopathic intracranial hypertension or IIH) is a condition where pressure builds up around the brain without an identifiable tumor — but the symptoms mimic a brain tumor.
Research suggested Mirena users were up to seven times more likely to develop pseudotumor cerebri. Symptoms include severe headaches, vision changes, balance problems, ringing in the ears, and in severe cases, permanent vision loss. About 200 lawsuits were filed alleging this link by 2017, and they were consolidated into a separate MDL — but that MDL was ultimately dismissed when judges excluded plaintiff experts.
What is an MDL and how is it different from a class action?
Quick Answer: An MDL (multidistrict litigation) consolidates many similar lawsuits under one judge to streamline pretrial proceedings, but each case remains individual — with its own facts, damages, and potential payout.
A class action, by contrast, groups all plaintiffs into one case with one settlement distributed among all members. Mirena’s main litigation was MDL-based, meaning each woman’s compensation was tied to her specific injuries. That’s why Mirena settlement amounts were not publicly announced as a per-person figure — they varied by case.
What is Bayer’s position on Mirena safety?
Quick Answer: Bayer has consistently defended Mirena as safe and effective when used as directed, attributing many reported complications to pre-existing conditions or insertion technique rather than the device itself.
Bayer updated Mirena’s labeling over the years to include additional warnings, and it has argued in court that it fully complied with FDA disclosure requirements. In the 2022 breast cancer class action, Bayer claimed its label already disclosed levonorgestrel as a hormone and that the breast cancer risk was a known, disclosed class effect of hormonal contraceptives.
How do I know if my Mirena caused organ perforation?
Quick Answer: Symptoms of Mirena migration or perforation include sudden or worsening pelvic pain, pain during intercourse, inability to feel the device’s strings, and in some cases, abdominal pain spreading beyond the uterus.
Perforation is typically confirmed through imaging — an ultrasound, X-ray, or CT scan can show whether the device has moved from its intended position. Some perforations are discovered during removal procedures when a doctor can’t locate the device in the uterus. If surgery was required to remove your Mirena, this is a strong indicator that migration occurred. LOLA Tampons Lawsuit 2026
What happened to the 2009 FDA warning letter to Bayer?
Quick Answer: In 2009, the FDA sent Bayer a formal warning letter citing deceptive and misleading promotion of Mirena at in-home marketing events where the company promised the device would improve relationships and sex lives — while downplaying real risks.
The 2009 FDA warning letter became significant evidence in Mirena lawsuits, showing that regulators had flagged Bayer’s marketing practices over a decade before the MDLs concluded. It supported plaintiffs’ arguments that Bayer prioritized sales messaging over accurate risk disclosure.
If I used Mirena and had breast cancer, what should I do right now?
Quick Answer: Contact a mass tort or product liability attorney immediately for a free consultation. Gather all your medical records — including Mirena insertion/removal records and all cancer diagnosis and treatment records.
The October 2024 Danish research is the strongest scientific evidence yet linking levonorgestrel IUDs to breast cancer. Attorneys are evaluating whether new cases are viable based on this updated science. Time matters because statutes of limitations are running — but the discovery rule may give you more time than you think if you only recently connected your cancer to Mirena use.
What is the statute of limitations for Mirena lawsuits?
Quick Answer: It varies by state, but most product liability statutes of limitations run 2 to 4 years from the date of injury or discovery of the injury’s cause.
The “discovery rule” is important here: your clock may not have started until you knew (or reasonably should have known) that Mirena caused your injury. If you only recently read about the breast cancer research and connected it to your diagnosis, you may still be within the window. Some states also have a separate statute of repose — a hard deadline regardless of discovery — so consulting an attorney quickly is essential.
What other birth control devices are currently being litigated?
Quick Answer: The most active birth control device lawsuits as of 2026 are the Paragard IUD lawsuit (broken device during removal; ~3,800 active cases) and the Depo-Provera lawsuit (brain tumors/meningiomas; 1,470+ cases).
Women harmed by those devices have active MDLs with bellwether trials underway or scheduled. If you experienced injuries from Paragard or received Depo-Provera injections and were later diagnosed with a meningioma, those cases are being actively accepted by attorneys.
Can I opt out of a Mirena settlement?
Quick Answer: The prior Mirena MDL settlements are closed, so opting out is no longer possible for those proceedings. If a new class action settlement is announced in the future, you would have an opt-out window at that time.
In any class action or MDL settlement, you typically have the right to opt out and pursue your own individual lawsuit instead. This makes sense if your damages are unusually high and an individual case might recover more. Opting out requires meeting a specific deadline stated in the settlement notice.
Where can I find more information about the Mirena lawsuits?
Quick Answer: Court documents for MDL 2434 (perforation cases) and the New York pseudotumor cerebri MDL are available through PACER (Public Access to Court Electronic Records) at pacer.gov. The breast cancer class action (Case No. 5:22-cv-01603) is accessible through the N.D. California PACER portal.
Drugwatch.com, AboutLawsuits.com, and Lawsuit-Information-Center.com maintain updated coverage of ongoing developments. For legal advice specific to your situation, consult a licensed attorney in your state.
A Note on Related Litigation Worth Watching
The Mirena litigation ended largely unfavorably for plaintiffs — but IUD and hormonal birth control litigation is far from over. Women harmed by the Paragard IUD (which breaks during removal, leaving fragments inside the body) are part of a growing MDL with nearly 3,800 pending cases as of January 2026, with bellwether trials scheduled. Women who developed brain tumors (meningiomas) after Depo-Provera injections have an active and growing MDL with over 1,470 cases — and the FDA updated Depo-Provera’s label in December 2025 to include brain tumor risk.
If you used Mirena and it worked without complications, the existing lawsuits don’t affect you. But if you’re among the tens of thousands who reported serious problems — and especially if you later developed breast cancer — the legal picture in 2026 is still evolving. New science, new attorneys, and potentially new litigation could emerge.
The best thing you can do right now is consult an attorney, preserve your records, and stay informed.
