The Phoenix ED device lawsuit is an active class action filed against the manufacturer of a widely marketed at-home erectile dysfunction treatment device, with plaintiffs claiming they were misled by false advertising, a fraudulent FDA approval claim, undisclosed side effects, and a refund policy the company allegedly never honored. The case was filed in August 2022 in a California state court and is currently progressing through discovery and class certification. No final settlement has been reached yet — but if you bought this device and felt misled, you may be able to join the class action and seek compensation.
Quick Answer The Phoenix ED device lawsuit, filed in August 2022, targets Nutraceutical Corporation over claims of false advertising, no real FDA approval, undisclosed side effects, and refused refunds. Plaintiffs are seeking at least $5 million in damages. The case is still active in California state court. There is no claim deadline yet — but you should contact an attorney now to protect your rights before statutes of limitations run out.
If you or someone you know bought the Phoenix ED device and experienced disappointing results, side effects like blisters or discomfort, or had a refund denied, this guide breaks down exactly what happened, who can join the lawsuit, what compensation might look like, and what steps to take right now. National Life Group Lawsuit

What Is the Phoenix ED Device Lawsuit About?
Background of the Lawsuit
The Phoenix is a handheld device marketed directly to men as a non-invasive, at-home treatment for erectile dysfunction. It uses a technology called low-intensity extracorporeal shockwave therapy (Li-ESWT) — the idea being that acoustic waves stimulate blood flow to penile tissue and help restore natural erectile function, without pills, injections, or surgery.
The device retails for several hundred dollars, and the company behind it, Nutraceutical Corporation (operating through its brand Launch Medical), marketed it heavily online with claims of clinical proof, high success rates, and — critically — FDA approval. The problem? Those claims appear not to hold up to scrutiny, according to the consumers who sued.
When buyers discovered the device wasn’t FDA-cleared for treating ED, didn’t work as advertised, caused painful side effects in some cases, and came with a “risk-free guarantee” the company often refused to honor, a group of them took legal action. Their class action lawsuit, filed in California, alleges that Nutraceutical ran a deceptive marketing campaign that misled thousands of men seeking help for a sensitive and personal health condition.
Timeline of Key Events
| Date | Event | Details |
|---|---|---|
| 2019–2021 | Phoenix device gains popularity | Nutraceutical/Launch Medical aggressively markets the Phoenix online as a clinically proven, FDA-backed home ED treatment |
| Early 2022 | Consumer complaints escalate | Buyers report ineffective results, side effects, and denied refund requests despite the advertised 60-day guarantee |
| August 2022 | Class action lawsuit filed | Peiffer Wolf Carr Kane & Conway files suit in California state court against Nutraceutical Corporation and CEO Dwayne Gutierrez |
| November 2022 | Arbitration bid denied | Nutraceutical attempts to move the case out of court to private arbitration — the California judge refuses, keeping the case in open state court |
| 2023 | Discovery and investigation phase | Plaintiffs begin gathering internal documents, marketing materials, and customer complaint records |
| 2023–2024 | Separate GAINSWave dispute resolved | A related lawsuit between Launch Medical and GAINSWave (a competing shockwave therapy provider) is settled, with the two companies agreeing to divide the market |
| 2024–2025 | Class certification proceedings | Plaintiffs pursue class certification to allow all affected buyers to be part of the case |
| 2025–2026 | Case remains active | No final settlement reached; case continues through California courts with $5 million in damages sought |
| TBD | Potential settlement or trial | Settlement talks or a full trial date have not yet been announced publicly |
Who Filed the Lawsuit?
The case was brought by a group of dissatisfied Phoenix device purchasers and is being handled by the law firm Peiffer Wolf Carr Kane & Conway, a well-known consumer rights and class action firm. The defendants are Nutraceutical Corporation — the company that manufactures and markets the Phoenix — and its CEO, Dwayne Gutierrez. The case was filed in California state court, where consumer protection laws are among the strongest in the country.
What Are the Allegations?
The lawsuit doesn’t pull punches. Here’s what plaintiffs are specifically claiming:
- ✅ False advertising about effectiveness — Nutraceutical advertised the Phoenix as a clinically proven treatment with high success rates. Plaintiffs say there is no adequate scientific evidence supporting those claims for home use of this specific device.
- ✅ Fraudulent FDA approval claims — The company allegedly implied or directly stated the device was FDA-approved for treating ED. It is not. This is one of the most serious allegations in the case, since FDA approval is a major trust signal for consumers buying health devices.
- ✅ Failure to disclose side effects — Some users experienced itching, blisters, skin discoloration, and penile discomfort. Plaintiffs claim the company never adequately warned them these risks existed, and that the device’s instructions were vague about safe use.
- ✅ Fraudulent refund policy — Nutraceutical prominently advertised a 60-day “risk-free guarantee.” But when buyers tried to return the device, many say their requests were denied or ignored — leaving them stuck with an expensive device they didn’t want and didn’t find effective.
Who Qualifies to Join the Phoenix ED Device Lawsuit?
Quick Answer You may be eligible to join the lawsuit if you purchased a Phoenix ED device and were misled by its advertising, experienced side effects, or had a legitimate refund request denied. Because the case is still in class certification, the exact criteria are still being established — but contacting an attorney now is the smartest move.
Eligibility Requirements
Because the Phoenix ED device lawsuit hasn’t yet been formally certified as a class action or reached settlement, there’s no official claim form open right now. But the likely criteria for joining the class are based on what the plaintiffs are alleging:
| Requirement | Details | Documentation Needed |
|---|---|---|
| You purchased the Phoenix ED device | You must have bought the product directly from the company or through an authorized channel | Order confirmation, bank/credit card statement, email receipt |
| You were in the US at the time of purchase | The lawsuit is filed in California but likely covers US buyers broadly | Billing address on order confirmation |
| You relied on marketing claims | You purchased partly based on claims about FDA approval, clinical proof, or guaranteed results | Screenshots of ads you saw, marketing materials |
| You experienced harm | Device didn’t work as promised, you suffered side effects, or a valid refund was denied | Medical records (if applicable), written refund request records, device photos |
| Your purchase is within the statute of limitations | California typically has a 4-year statute for fraud and consumer protection claims | Purchase date on your receipt or bank records |
Eligible Device — The Phoenix (Li-ESWT Home Device)
| Device | Manufacturer | Marketed As | Issue |
|---|---|---|---|
| The Phoenix | Nutraceutical Corporation / Launch Medical | At-home Li-ESWT device for erectile dysfunction | Alleged false FDA claims, misleading efficacy claims, undisclosed side effects |
This lawsuit specifically targets the Phoenix home-use device marketed by Nutraceutical. Note that a separate professional-grade version operated through licensed GAINSWave clinics is a different product — that dispute was separately resolved between the two companies in 2023. If you used the device through a clinic, your situation may differ; consult an attorney.
Who Does NOT Qualify?
- ❌ People who purchased outside the United States
- ❌ People whose purchase falls outside the statute of limitations (generally more than 4 years ago, depending on your state)
- ❌ People who received the device as a free promotional sample with no purchase
- ❌ People who already settled individual claims against Nutraceutical separately
How to Document Your Claim Right Now
Even before there’s a formal claim form, collecting your evidence now is critical. Here’s what to pull together:
| Document | Why It Matters | Where to Find It | If You Don’t Have It |
|---|---|---|---|
| Purchase receipt or order confirmation | Proves you bought the product | Email inbox, company website order history | Check bank/credit card statements for the charge |
| Marketing materials or ads you saw | Shows what claims you relied on | Screenshot web archive (web.archive.org) or your email promotions folder | Describe the claims from memory in writing |
| Refund request records | Proves the company denied a valid return | Emails to/from company support | Check your sent mail folder or live chat transcripts |
| Medical records (if side effects occurred) | Documents physical harm | Your doctor or urgent care provider | Write a detailed account of symptoms with dates |
| Photos of the device or packaging | Shows FDA claims or labeling | Take photos if you still have the device | Look for photos in your email confirmation |
How Much Compensation Could You Get?
Quick Answer No settlement has been finalized yet, so there are no confirmed payout amounts. Plaintiffs are seeking at least $5 million in total damages. If the case settles, compensation could include full or partial refunds of the device purchase price, additional damages for side effects, and potentially punitive damages. Lawsuit in Spanish
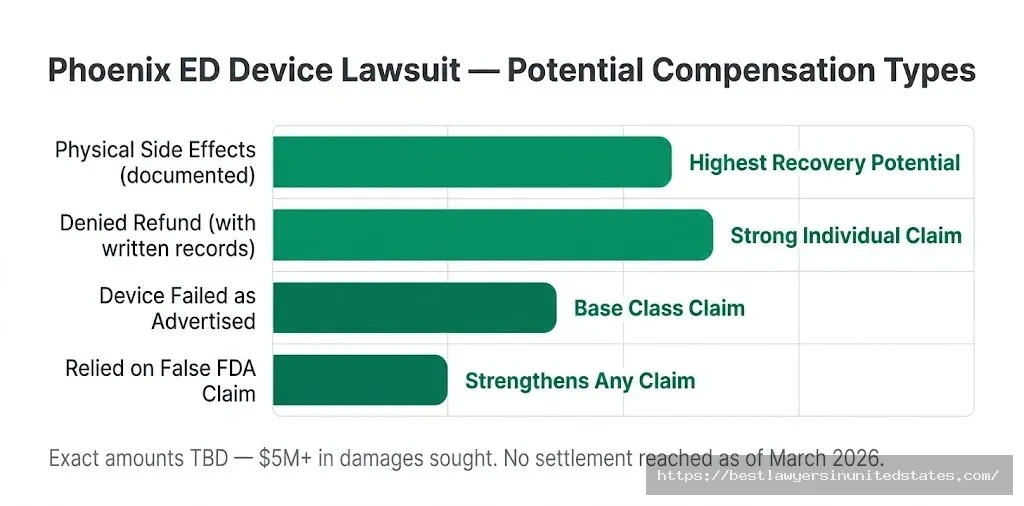
Damages Sought in the Lawsuit
| Category | Details | Status |
|---|---|---|
| Total damages sought by plaintiffs | At least $5 million | Claimed — not yet awarded |
| Consumer refunds | Return of the purchase price (device typically sold for several hundred dollars) | Potential outcome if settlement reached |
| Additional compensatory damages | Compensation for side effects, medical expenses, emotional distress | Possible depending on individual claims |
| Punitive damages | Additional penalties if court finds willful deception | Possible in California consumer fraud cases |
| Non-monetary relief | Changes to marketing language, labeling corrections, refund policy reforms | Likely included in any settlement terms |
What Factors Would Affect Your Individual Payout?
In a class action settlement, what you get typically depends on several factors:
- Whether you experienced physical side effects — Those who suffered documented harm like blisters or discomfort generally get higher compensation than those who simply found the device ineffective.
- The purchase price you paid — Refunds are often tied to what you actually spent, so keeping your receipt matters.
- Whether your refund was denied — If the company explicitly rejected a valid return request, that strengthens your individual claim.
- Number of total class members — The more people who join the class, the more it divides the fund — but it also raises pressure on the defendant to settle for more.
- Whether you can document reliance on the false FDA claim — This is a key allegation; showing you bought because of that claim adds weight to your case.
⚠️ Important: Because no settlement has been reached or announced, there are no official payout amounts to quote. Any website claiming exact settlement dollar figures for this case right now is speculating. This guide gives you accurate current information — the case is active and unresolved as of March 2026.
How to Join or File a Claim — Step by Step

⚠️ No Official Claim Deadline Yet — But Don’t Wait
There is no formal claim form open yet because the case hasn’t settled. However, statutes of limitations still apply. In California, you typically have 4 years to bring a fraud or consumer protection claim. If your purchase was in 2020 or earlier, your window may be closing. Contact an attorney now — it costs nothing for a consultation.
Step 1: Gather Your Documentation Pull together everything listed in the documentation table above. At a minimum, find your purchase receipt or bank statement showing the charge. The stronger your paper trail, the better your position. Don’t discard the device — photos of the packaging and any labeling showing FDA claims could be valuable evidence.
Step 2: Contact a Consumer Rights Attorney The case is handled by Peiffer Wolf Carr Kane & Conway, but you can also reach out to any consumer rights or class action attorney in your state. Most offer free consultations. They can tell you quickly whether your situation fits the class and advise you on the best path forward. You can also email admin@bestlawyersinunitedstates.com for a referral.
Step 3: Register Your Interest With the Plaintiffs’ Firm Law firms handling active class actions maintain lists of potential class members. Reaching out to Peiffer Wolf Carr Kane & Conway puts you on their radar — they can notify you the moment a claim form becomes available or a settlement is reached.
Step 4: Watch for Court Notices Once class certification is granted, the court requires that class members be notified — typically by email, mail, or published notice. If you bought through the official website, they may have your contact info already. Keep an eye on your inbox.
Step 5: File Your Claim Once Available When a settlement is reached and a claim form opens, you’ll need to submit your documentation, describe your experience with the device, and provide your contact and payment information. Missing that deadline once it’s announced could bar you from any recovery.
Step 6: Save Everything Keep all attorney communications, any claim confirmation numbers, and copies of whatever you submit. Class action settlements can take months to distribute after filing.
Step 7: Decide Whether to Opt Out When a settlement is announced, you’ll have the option to opt out and pursue an individual lawsuit if you believe your damages are significant enough to warrant it. Most people stay in the class — but if you suffered serious physical harm, talk to your attorney before the opt-out deadline.
Critical Dates to Watch
| Milestone | Status | What to Do |
|---|---|---|
| Lawsuit filed | August 2022 ✅ | N/A — already happened |
| Arbitration denied | November 2022 ✅ | N/A — already happened |
| Class certification decision | Pending — expected 2025/2026 | Follow news or attorney updates |
| Settlement announcement | Not yet announced | Sign up for legal alerts or attorney notification |
| Claim form deadline | TBD — announced after settlement | Watch for notices; contact attorney now |
| California fraud statute of limitations | 4 years from purchase date | If you bought before early 2022, contact an attorney immediately |
Current Lawsuit Status & 2026 Updates
Where Things Stand Right Now (March 2026)
The Phoenix ED device lawsuit remains active in California state court. As of early 2026, no settlement has been announced, and no claim filing portal has opened. The case is progressing through the pre-trial litigation process, which typically includes class certification hearings, discovery (exchanging evidence), and depositions.
A significant early win for plaintiffs came in late 2022, when the California court rejected Nutraceutical’s bid to push the case into private arbitration — a move that would have made it much harder for consumers to get justice as a group. That ruling kept the lawsuit in open court and on track for class action treatment.
Separately, a prior dispute between Launch Medical and GAINSWave — a competing clinical shockwave provider — was resolved in 2023. That settlement divided the market between the two companies (Launch Medical keeps home-use rights; GAINSWave handles professional clinical use) but did not address consumer claims about the Phoenix device’s effectiveness or marketing.
Recent Developments
- 2025: Legal analysts and consumer rights publications highlighted that the Phoenix lawsuit raises broader industry questions about FDA oversight of direct-to-consumer medical devices — signaling the case has attracted significant legal and regulatory attention beyond just the two parties.
- Early 2026: The case continues with discovery ongoing. No trial date has been publicly set, and settlement discussions have not been publicly confirmed.
- Ongoing: Consumer reviews of the Phoenix device remain mixed, with the device still available for purchase online despite the litigation — a situation legal analysts say highlights the gap in regulatory oversight of at-home health tech.
What to Expect Next
- Class certification ruling — The court will decide whether to formally certify the lawsuit as a class action, allowing all affected buyers to be treated as a group. This is a major milestone.
- Discovery completion — Internal documents from Nutraceutical about their marketing decisions, internal testing (if any), and customer complaint handling could become public.
- Settlement talks or trial — Most class actions of this type settle before trial. If Nutraceutical wants to avoid a public trial and the discovery of damaging internal records, settlement talks may begin soon.
- Regulatory action — The case has drawn attention to the FDA’s oversight (or lack thereof) of at-home wellness devices, and regulatory changes could follow.
The Phoenix ED Device Lawsuit vs. Similar Cases
How This Compares to Other Medical Device Class Actions
| Lawsuit | Defendant | Allegations | Damages Sought / Awarded | Status |
|---|---|---|---|---|
| Phoenix ED Device (this case) | Nutraceutical / Launch Medical | False advertising, fake FDA claims, denied refunds, undisclosed side effects | $5 million+ | Active — pending class certification |
| Hims & Hers (various) | Hims, Inc. | Subscription billing disputes, auto-renewal complaints | Varies by case | Multiple — various stages |
| iHealth Medical Device FDA Cases | Various manufacturers | Selling uncleared medical devices as FDA-approved | FTC penalties vary | FTC enforcement actions ongoing |
| NovaBay / Wound Care Device | Various | Misleading efficacy claims for home medical devices | Multi-million settlements | Settled |
What Makes This Lawsuit Stand Out
Most false advertising class actions target companies that exaggerate results. The Phoenix case adds a more serious layer: an alleged outright false claim of FDA approval. Telling consumers a medical device has FDA clearance when it doesn’t is not just a marketing gray area — it’s the kind of allegation that can lead to punitive damages, because it implies a knowing deception.
The fact that the California court already blocked arbitration is also significant. That ruling signals the judge took the plaintiffs’ case seriously enough to keep it in the public court system, where class actions can proceed and public records remain visible.
Do You Need a Lawyer to Join This Lawsuit?
Quick Answer You don’t need to hire your own attorney to eventually file a claim in this class action — once a settlement is reached, a simple claim form is usually all you need. But right now, while the case is still being litigated, consulting a lawyer can help you protect your rights, especially regarding statutes of limitations.
Filing Without a Lawyer (Once Settlement Is Reached)
When class action lawsuits settle, the claims process is generally designed to be straightforward. You’ll typically fill out an online or mail-in form, provide basic proof of purchase, describe what happened, and submit. Court-appointed attorneys handle the legal heavy lifting on behalf of the entire class — you don’t pay anything out of pocket. Attorney fees in class actions come out of the settlement fund, not from your individual payout. Chris Brown Lawsuit 2026
When Getting Legal Help Makes Sense Right Now
Even though you don’t technically need a personal attorney to eventually file a claim, there are good reasons to consult one before the case settles:
- If you suffered significant physical side effects and want to explore an individual (non-class) lawsuit for larger compensation
- If your purchase date is approaching the statute of limitations window
- If you’re unsure whether your specific experience qualifies
- If you want to understand your opt-out rights before the deadline arrives
Free Legal Consultation
The lead law firm on the case is Peiffer Wolf Carr Kane & Conway, which handles consumer class actions across the country. Most consumer rights attorneys offer free initial consultations and work on contingency — meaning you pay nothing unless you recover money. For a referral to a qualified attorney in your area, contact admin@bestlawyersinunitedstates.com.
Frequently Asked Questions
What is the Phoenix ED device lawsuit?
Quick Answer A class action lawsuit filed in August 2022 in California against Nutraceutical Corporation, the maker of the Phoenix at-home erectile dysfunction device, alleging false advertising, fake FDA approval claims, undisclosed side effects, and a fraudulent refund policy.
The lawsuit was brought by dissatisfied buyers who say they were deceived into paying several hundred dollars for a device that didn’t perform as advertised — and then couldn’t get their money back when they tried to use the company’s own guarantee.
Who is being sued?
Quick Answer Nutraceutical Corporation and its CEO, Dwayne Gutierrez, are the named defendants.
The company markets the Phoenix under its Launch Medical brand. The CEO is named individually because the lawsuit alleges knowing participation in deceptive marketing decisions.
Has the lawsuit been settled?
Quick Answer No. As of March 2026, no settlement has been reached or announced. The case is still working through California state court.
The lawsuit is in active litigation, with class certification pending. Settlement talks may occur privately, but nothing public has been confirmed.
Is there a claim form I can fill out right now?
Quick Answer No — there is no official claim form yet. Claim forms open after a settlement is reached and approved by the court.
What you can do right now is document your experience, gather your purchase records, and contact an attorney to register your interest so you don’t miss the filing window when it opens.
How much money could I get?
Quick Answer Unknown — no settlement amount has been established. Plaintiffs are seeking $5 million+ in total damages.
If and when the case settles, compensation would likely include refunds of the purchase price and potentially additional damages for those who experienced side effects. The exact per-person amount depends on the total number of claimants and the final settlement terms.
What is the filing deadline?
Quick Answer There’s no formal claim deadline yet — but California’s statute of limitations for fraud is 4 years from the date of purchase. If you bought in early 2022 or earlier, your window may be narrowing.
Once a settlement is announced, a specific claim deadline will be set — typically 60 to 180 days after settlement approval. Missing that deadline bars you from recovery. evan elan attorney
Do I need a lawyer?
Quick Answer Not necessarily — but consulting one now is smart. You don’t need personal legal representation to file a claim once a settlement is open. But an attorney can help you protect your rights before the deadline.
Who qualifies to join the lawsuit?
Quick Answer Generally, anyone in the US who purchased the Phoenix device and was misled by its marketing, experienced side effects, or had a refund wrongly denied.
Class membership will be formally defined when the court certifies the class. The criteria will likely focus on US purchases made within the limitations period from buyers who relied on the company’s advertising claims.
What if I don’t have my receipt?
Quick Answer You may still qualify. Bank or credit card statements showing the charge, email confirmations, or even records of your refund request can serve as proof of purchase.
In most class action settlements, strict receipt requirements are loosened for buyers who make a good-faith effort to document their purchase through available records.
Is the Phoenix device still being sold?
Quick Answer Yes. Despite the active lawsuit, the Phoenix is still available for purchase online as of early 2026.
This is not unusual — products can remain on the market while litigation proceeds. It does highlight, however, that the FDA has limited ability to immediately pull non-prescription health devices from sale during a civil lawsuit. If you’re considering buying it, speak with a doctor first.
What is Li-ESWT, and does it actually work?
Quick Answer Low-intensity extracorporeal shockwave therapy does have some support in clinical settings, but evidence for at-home use of devices like the Phoenix is limited and disputed.
The core of the lawsuit isn’t necessarily that shockwave therapy never works — it’s that this specific device, used at home without clinical supervision, was marketed with exaggerated claims and without the FDA clearance the company implied it had. Clinical settings use calibrated equipment with trained practitioners; home devices are a different matter.
What side effects did people report?
Quick Answer Plaintiffs reported itching, blisters, skin discoloration, penile discomfort, and pain after use.
The lawsuit alleges these risks weren’t adequately disclosed and that the device’s instructions were vague, making safe home use difficult. If you experienced any of these effects, document them and speak with a doctor and attorney.
What was the GAINSWave lawsuit about — is it the same case?
Quick Answer No. The GAINSWave dispute was a separate business-to-business lawsuit between Launch Medical and a competing therapy provider. That case settled in 2023. The consumer class action is an entirely different case.
The GAINSWave settlement resolved a market competition dispute and allowed the two companies to divide commercial territory. It did nothing to resolve the consumer fraud claims in this lawsuit.
Can I sue separately instead of joining the class action?
Quick Answer Yes — you have the right to opt out of the class action and file your own individual lawsuit. This is usually only worth it if your damages are unusually high.
Most buyers opt into the class because individual lawsuits are expensive and slow. But if you suffered serious documented physical harm, an individual claim may produce higher compensation. Talk to an attorney before the opt-out deadline — which hasn’t been set yet.
What if I already got a partial refund from the company?
Quick Answer You may still qualify, but your claim amount might be reduced by the amount you already received. Consult an attorney to understand your specific situation.
Will compensation from this lawsuit be taxable?
Quick Answer It depends on the type of compensation. Refunds for the purchase price are generally not taxable. Damages for emotional distress, punitive damages, or other non-physical harm may be taxable income.
Talk to a tax professional once you receive any settlement payment. The settlement documentation may also clarify how the IRS should treat different payment categories.
How will I know when a settlement is reached?
Quick Answer The court requires class members to be notified — usually by email, postal mail, or a published legal notice. You can also monitor news about the case or sign up for alerts with the plaintiffs’ law firm.
What happens if the lawsuit is dismissed?
Quick Answer If the case is dismissed or Nutraceutical wins, there would be no class action settlement and no claim form. You’d still have the option to pursue an individual lawsuit within the limitations period.
Given that the court already rejected the defendant’s attempt to move to arbitration, dismissal is not the most likely outcome — but it remains a possibility in any litigation.
What precedent could this case set?
Quick Answer Legal experts say this case could reshape how at-home medical devices are marketed and regulated — particularly around FDA approval claims and refund guarantee enforcement.
If the court rules against Nutraceutical, it could signal to the broader at-home health device industry that inflated claims and fake regulatory endorsements carry real legal risk. It may also prompt the FDA to increase oversight of this growing product category. 307 ipc in bns
Bottom Line: What Should You Do Right Now?
If you bought the Phoenix ED device and felt misled, experienced side effects, or had a refund denied, here’s your action plan for today:
- Gather your purchase records — receipt, bank statement, email confirmation
- Document any side effects — in writing, with dates, and get medical records if you saw a doctor
- Save any refund request records — emails, chat logs, letters from the company
- Contact an attorney — for a free consultation to understand if you’re within the statute of limitations and what your options are
- Watch for court notices — especially if the company has your email address from the original purchase
- Bookmark this page — we’ll update as the case develops, class certification is decided, and settlement news breaks
This guide will be updated as the case progresses. For attorney referrals, email admin@bestlawyersinunitedstates.com or consult a consumer rights attorney in your state.
Last updated: March 2026. This article is for informational purposes only and does not constitute legal advice. For advice specific to your situation, consult a licensed attorney. Information sourced from court filings, legal news coverage, and consumer protection reporting.
