The Tepezza lawsuit involves hundreds of people who received Tepezza infusions for thyroid eye disease and later developed permanent hearing loss or tinnitus. Unlike a typical class action, these are individual claims consolidated into a federal multidistrict litigation (MDL) — which means there is no simple claim form to fill out. Instead, you need to contact an attorney and file your own lawsuit. As of February 2026, no global settlement has been announced, but trial deadlines have been struck and settlement talks are actively underway, suggesting a resolution may be near. Zicam Lawsuit: 2026 Complete Guide
Quick Answer: Tepezza (teprotumumab-trbw), manufactured by Horizon Therapeutics, has been linked to permanent hearing loss and tinnitus in patients treated for thyroid eye disease. More than 273 lawsuits are currently pending in federal MDL in Illinois. No settlement has been finalized yet, but attorney estimates place individual case values at $75,000 to $500,000+ depending on injury severity. If you received Tepezza infusions and experienced hearing problems, contact a lawyer now — state statute of limitations deadlines (typically 2–3 years from injury) may be approaching or already passed for some patients. Facial abuse lawsuit

What Is the Tepezza Lawsuit About?
Background of the Lawsuit
Tepezza (generic name: teprotumumab-trbw) was approved by the FDA in January 2020, making it the first and only drug specifically approved to treat thyroid eye disease (TED). TED is a rare autoimmune condition that causes inflammation behind the eyes, resulting in painful symptoms including bulging eyes, double vision, and potential vision loss. For patients with TED, Tepezza was marketed as a breakthrough — and for the eyes, it largely worked.
The problem was what it was doing to patients’ ears.
Shortly after Tepezza hit the market, patients and their doctors began noticing something alarming: a significant number of people receiving Tepezza infusions were developing hearing problems. These ranged from ringing in the ears (tinnitus) to muffled hearing to permanent, irreversible hearing loss. Independent studies found the drug’s actual hearing risk was far higher than what Horizon Therapeutics disclosed to patients or the FDA during clinical trials.
At the heart of every Tepezza lawsuit is a simple allegation: Horizon Therapeutics knew — or should have known — about these hearing risks, and failed to warn patients or doctors in time to prevent permanent injuries. The company did not update Tepezza’s drug label to include hearing loss warnings until July 2023, more than three years after the drug launched.
Timeline of Key Events
| Date | Event | Details |
|---|---|---|
| January 2020 | FDA approves Tepezza | First drug approved for thyroid eye disease |
| Early 2021 | Patient hearing complaints emerge | Reports of tinnitus and hearing loss begin accumulating |
| March 2021 | Endocrine Society study published | Found 65% of patients with 4+ infusions developed hearing problems |
| February 2022 | American Journal of Ophthalmology study | Found 81.5% of 27 patients developed new or worsening hearing symptoms |
| Fall 2022 | First Tepezza lawsuit filed | Product liability claim in federal court |
| July 2023 | FDA updates Tepezza label | Adds warnings for permanent hearing loss; recommends audiological monitoring |
| June 2023 | MDL established | U.S. JPML consolidates all federal cases before Judge Thomas Durkin in Northern District of Illinois |
| November 2023 | Bellwether program created | Court selects representative cases for early trials |
| June 2024 | Bellwether pool set | 12 cases designated for bellwether discovery |
| October 2025 | All trial deadlines struck | Court removes bellwether deadlines, fueling settlement speculation |
| January 8, 2026 | Status conference held | Court and parties discuss case management and next steps |
| August 3, 2026 | First bellwether trial (if no settlement) | Rescheduled from earlier dates |
Who Filed the Lawsuit?
The Tepezza lawsuits are not a class action — they are individual product liability claims filed by patients who received Tepezza infusions and then suffered hearing damage. As of January 2026, there are 273 active cases pending in the MDL, with 282 total cases filed. Cases come from patients across all 50 states.
Major law firms currently representing Tepezza plaintiffs include Miller & Zois, TorHoerman Law, Lanier Law Firm, Seeger Weiss LLP, and Weitz & Luxenberg, among others. The defendant is Horizon Therapeutics (now a subsidiary of Amgen, which acquired Horizon in 2023).
The MDL is overseen by U.S. District Judge Thomas Durkin in the Northern District of Illinois (Chicago).
What Are the Allegations?
Every Tepezza lawsuit rests on similar core claims. Patients allege that Horizon Therapeutics:
- Failed to warn patients and doctors that Tepezza could cause permanent hearing loss, despite evidence from clinical trials and independent studies that the risk existed
- Concealed or downplayed internal data showing the true rate of hearing-related adverse events — including data from its own Study 401, which showed up to 40% of patients experienced auditory problems
- Delayed updating the drug label by years, even as post-marketing data and independent research confirmed the link between Tepezza and serious hearing damage
- Did not recommend audiological monitoring before, during, or after treatment — a precaution that might have caught hearing damage earlier and allowed patients to stop treatment before permanent injury occurred
- Misrepresented the drug’s safety profile to the FDA during the approval process
The consequence for patients: by the time they realized Tepezza was hurting their hearing, the damage was often already permanent and irreversible.
Who Qualifies for a Tepezza Lawsuit?
Quick Answer: You may qualify if you received Tepezza infusions for thyroid eye disease and later developed hearing loss, tinnitus, muffled hearing, or other auditory problems — especially if your symptoms appeared before July 2023 when warning labels were finally updated. You’ll need medical records documenting both your Tepezza treatment and your hearing diagnosis. Contact an attorney before your state’s statute of limitations expires (usually 2–3 years from injury or discovery of injury).
Eligibility Requirements
| Requirement | Details | Documentation Needed |
|---|---|---|
| Received Tepezza infusions | Any number of infusions for thyroid eye disease or Graves’ disease | Medical records showing Tepezza prescriptions and infusion dates |
| Developed hearing-related symptoms | Hearing loss, tinnitus, muffled hearing, autophony, Eustachian tube dysfunction, or balance problems | Audiological evaluation, ENT records, diagnosis records |
| Symptoms linked to Tepezza use | Hearing problems appeared during or after infusions | Timeline in medical records connecting treatment to onset of symptoms |
| Within statute of limitations | Generally 2–3 years from injury or discovery (varies by state) | Date of first hearing symptoms; date of Tepezza treatment |
Two or more infusions tend to produce stronger cases, since studies show hearing risk increases with more infusions. However, even patients who received only a few infusions have filed lawsuits.
Symptoms That Qualify
If you experienced any of these after Tepezza infusions, you may have a valid claim:
- Permanent sensorineural hearing loss (damage to inner ear hair cells)
- Tinnitus (persistent ringing, buzzing, or hissing in the ears)
- Muffled or distorted hearing
- Difficulty understanding conversations, especially in noisy environments
- Autophony (your own voice sounds too loud or echoes)
- Eustachian tube dysfunction
- Feeling of ear fullness or plugging
- Balance problems related to inner ear damage
- Patulous Eustachian tube (tubes stay open, causing constant sound distortion)
Who Does NOT Qualify?
You likely do not qualify if:
- You received Tepezza but have no hearing-related symptoms whatsoever
- Your hearing problems have been fully resolved with no lasting damage
- Your hearing loss has a documented unrelated cause (e.g., genetic condition, prior injury)
- Your statute of limitations has expired — this varies by state and depends on when you received treatment and when you first noticed symptoms (consult an attorney to confirm)
- You received Tepezza after July 2023 when warning labels were updated, though some attorneys may still take these cases depending on circumstances
How to Prove Your Claim
| Document Type | Why It’s Needed | Where to Find It | If You Don’t Have It |
|---|---|---|---|
| Tepezza infusion records | Proves you received the drug and when | Hospital or infusion center records, prescribing doctor’s office | Request from healthcare provider; insurance records may confirm |
| Audiological evaluation | Documents hearing loss or tinnitus diagnosis | ENT doctor, audiologist | Schedule an evaluation now — it’s important to get documented |
| Medical records showing symptom onset | Connects hearing problems to Tepezza timeline | Your primary care, ENT, or specialist records | Medical notes mentioning new hearing symptoms during treatment |
| Prescription records | Confirms Tepezza was prescribed and dispensed | Pharmacy records, insurance Explanation of Benefits | Insurance company or pharmacy can provide |
| Specialist referral records | Shows escalating care for hearing problems | Specialist’s office | Contact your referring physician |
How Much Money Could You Get?
Quick Answer: No Tepezza settlement has been finalized, so there are no guaranteed payout amounts. Attorneys estimate individual case values at $75,000 to $500,000+ for serious cases, with some estimates reaching higher for catastrophic permanent hearing loss. The final amounts will depend heavily on bellwether trial outcomes and settlement negotiations.
Why There Are No Confirmed Numbers Yet
This is critically important to understand: the Tepezza MDL has not settled. Unlike a class action settlement where you fill out a form and wait for a check, this litigation is still in active pretrial proceedings. The striking of bellwether trial deadlines in October 2025 suggests settlement talks are progressing — but no deal has been announced publicly as of February 2026.
The estimates below are based on attorney projections informed by similar pharmaceutical injury cases and the severity of injuries seen in this litigation. They are not guarantees. Spectrum Lawsuit 2026
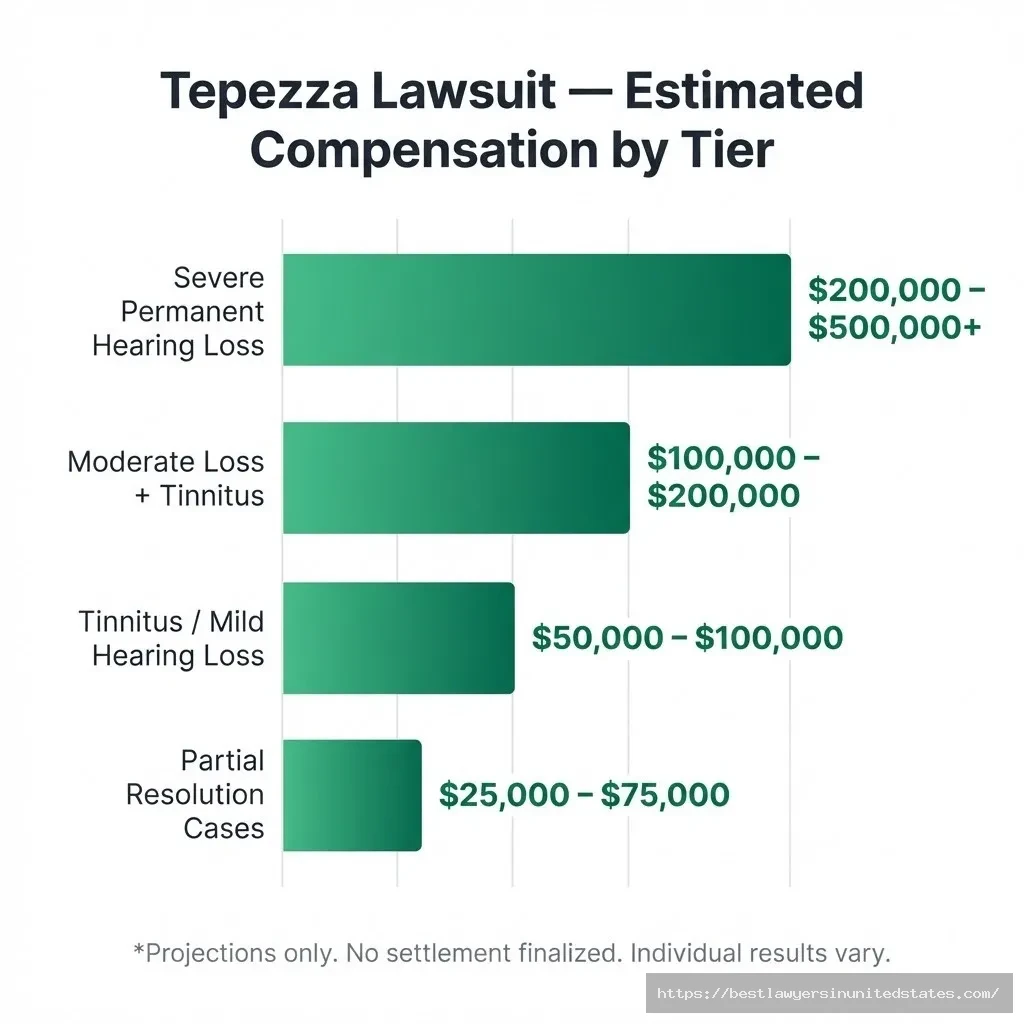
Projected Payout Tiers by Injury Severity
| Claim Category | Estimated Compensation Range | What Typically Qualifies | Evidence Strength Needed |
|---|---|---|---|
| Tier 1 – Severe Permanent Hearing Loss | $200,000 – $500,000+ | Documented permanent, irreversible hearing loss with major lifestyle impact; may require hearing aids or cochlear implants | Strong medical documentation; objective audiological testing |
| Tier 2 – Moderate Hearing Loss + Tinnitus | $100,000 – $200,000 | Significant but partial hearing loss combined with persistent tinnitus; measurable impact on daily life | Medical records + audiological evaluation |
| Tier 3 – Tinnitus or Mild Hearing Loss | $50,000 – $100,000 | Persistent tinnitus or mild-to-moderate hearing loss with documented onset after Tepezza | Medical diagnosis and clear timeline |
| Tier 4 – Partial Resolution Cases | $25,000 – $75,000 | Some improvement after stopping treatment, but not full recovery | Documentation of symptoms and partial resolution |
One estimate from Miller & Zois places favorable cases between $140,000 and $500,000. TorHoerman Law estimates $75,000 to $200,000+ as a general range. Individual outcomes will vary significantly.
What Affects Your Compensation
Your potential payout will depend on several factors:
Severity of the injury — Permanent, total hearing loss carries far more value than mild or resolved tinnitus. Cases where patients need hearing aids or cochlear implants after Tepezza are among the highest-value claims.
Evidence of causation — How clearly your medical records connect Tepezza treatment to the onset of your hearing symptoms matters enormously. If your audiological records show normal hearing before Tepezza and documented loss after, your case is stronger.
Number of infusions received — Studies show hearing risk increases with more infusions. Patients who received the full 8-infusion course before the 2023 label update may have stronger claims.
Treatment timeline — People treated before July 2023 (when warning labels were finally added) have stronger “failure to warn” claims. Your doctor had no label guidance during that time.
Impact on your life — Lost wages, inability to work in your field, mental health effects, and reduced quality of life all factor into damages.
How to Get Into the Tepezza Litigation
⚠️ IMPORTANT: This is NOT a class action. You cannot simply fill out an online claim form. To participate in this litigation, you need to hire an attorney who will file a product liability lawsuit on your behalf. Most Tepezza attorneys work on contingency — meaning you pay nothing unless they win.

Step-by-Step: How to Join the Tepezza MDL
Step 1: Contact a Tepezza attorney for a free consultation
Most law firms handling Tepezza cases offer free, no-obligation case reviews. You’ll describe your Tepezza treatment history and hearing symptoms. The attorney will tell you whether they think you have a viable case. This costs you nothing.
Step 2: Gather your medical records
Your attorney will help you obtain records, but you can start pulling these together:
- Tepezza infusion dates and medical records
- Audiological evaluations or ENT records
- Any records documenting hearing symptoms
Step 3: Your attorney files your lawsuit
If your attorney takes your case, they’ll file an individual product liability lawsuit against Horizon Therapeutics. Because the MDL exists, your case will be transferred to Judge Durkin’s court in the Northern District of Illinois for coordinated pretrial proceedings.
Step 4: Discovery and expert review
Your attorney handles the legal process — depositions, expert witnesses, document review. Your main job is to provide accurate medical history and cooperate with requests for information.
Step 5: Settlement or trial
If a global settlement is reached (which many attorneys expect will happen), your individual case will be valued within that framework. If no global settlement is reached, individual cases may go to trial or be remanded to home district courts.
Step 6: Receive compensation
In a settlement scenario, your attorney negotiates your individual share. They receive their contingency fee (typically 33–40%) from the settlement amount. You receive the remainder.
Critical Deadlines — State Statutes of Limitations
Unlike a class action with a single opt-in deadline, your Tepezza lawsuit must be filed before your state’s statute of limitations expires. This is the most important deadline to know about.
| State Group | Typical Timeframe | Clock Starts From |
|---|---|---|
| Most states | 2–3 years | Date of injury OR date you discovered (or should have discovered) the injury |
| Some states | 1 year | Date of injury |
| Discovery rule states | Extended | Allows filing from the date you linked symptoms to Tepezza |
If you received Tepezza infusions in 2020 or 2021 and noticed hearing problems shortly after, your deadline may have already passed or be approaching soon. Contact an attorney immediately — waiting is the single biggest mistake Tepezza victims make.
| What You Should Do | Timeframe |
|---|---|
| Contact an attorney for free consultation | Now — do not delay |
| Gather medical records and infusion history | Within days of consultation |
| Attorney files your lawsuit | As quickly as possible given your state deadline |
| Your case joins the MDL | Shortly after filing |
Common Mistakes to Avoid
Waiting to see if a settlement gets announced first — By the time a settlement is announced, your statute of limitations may have passed. You must file before deadlines, not after a deal is done.
Assuming this works like a class action — There’s no claim form, no website to visit and submit your information as a claimant. You need an attorney.
Not getting a hearing evaluation — If you haven’t had your hearing formally evaluated since your Tepezza treatment, do it now. Objective audiological data is critical to your case.
Using a law firm with no pharmaceutical MDL experience — Tepezza is complex litigation. Work with attorneys who have handled mass tort or pharmaceutical injury cases.
Current Lawsuit Status — February 2026 Update
Where Things Stand Right Now
The federal judge overseeing the Tepezza hearing loss multidistrict litigation removed all upcoming bellwether trial deadlines in October 2025, prompting speculation that Horizon Therapeutics may be nearing a global settlement. A status conference was scheduled for January 8, 2026, to address case management and potential next steps.
As of February 2026, approximately 273 active cases remain pending in the MDL. The dramatic move to strike all trial deadlines — without any rulings dismissing claims — is widely interpreted by legal observers as a signal that settlement talks are progressing seriously. When courts remove trial dates in mass tort litigation, it typically means both sides are engaged in active negotiations and don’t want trial preparation costs to derail the talks.
Judge Durkin has directed quarterly settlement discussions in an effort to resolve the litigation before trial, following nearly a year of required settlement negotiations.
Most Recent Developments
January 2026: The MDL held a status conference on January 8, 2026. Case count remained stable at 273 pending cases — no new filings in December or January.
October 2025: Judge Weisman held an off-the-record discussion with both parties’ counsel and struck all existing bellwether deadlines. This move fueled widespread speculation that a global resolution is close.
August 2025: The first Tepezza hearing loss bellwether trial was rescheduled to August 3, 2026, moving it back nearly two months. Additional bellwether cases are scheduled for September 28, 2026, November 30, 2026, and February 1, 2027. Those dates now appear moot given the striking of deadlines.
July 2025: Expert discovery concluded and both sides submitted expert reports, a key pretrial milestone.
What Happens Next
The most likely path forward is one of two scenarios:
Scenario 1 — Global Settlement (Most Likely): Horizon Therapeutics and plaintiffs’ attorneys reach a global resolution covering all or most of the 273+ pending cases. Individual case values would be negotiated within a settlement framework. Attorneys would then work with each plaintiff to accept or reject their individual allocation.
Scenario 2 — Bellwether Trials Proceed: If settlement talks break down, bellwether trials would resume. The outcomes of 2–3 trials would give both sides a clearer picture of jury sentiment and likely drive a post-trial settlement.
Tepezza Lawsuit vs. Similar Drug Injury Cases
How This Litigation Compares
| Lawsuit | Drug/Device | Injury | Status | Estimated Per-Person Range |
|---|---|---|---|---|
| Tepezza (Current) | Tepezza (teprotumumab) | Permanent hearing loss, tinnitus | Active MDL, ~273 cases, settlement likely | $75,000 – $500,000+ |
| Elmiron Lawsuit | Elmiron (pentosan polysulfate) | Vision damage, macular degeneration | Settled/ongoing | $50,000 – $300,000+ |
| Taxotere Lawsuit | Taxotere (docetaxel) | Permanent hair loss | Ongoing MDL | Varies significantly |
| Zofran Lawsuit | Zofran (ondansetron) | Birth defects | Largely dismissed | N/A |
| NEC Baby Formula | Cow-milk formula | Infant NEC disease | Active MDL | Varies by severity |
What Makes Tepezza Unique
A few things set the Tepezza litigation apart from other drug injury cases:
The science is unusually strong. Independent studies — including a 2021 Endocrine Society study and a 2022 American Journal of Ophthalmology study — found hearing problem rates of 65% and 81.5% respectively among Tepezza users, compared to just 10% disclosed during clinical trials. That gap is extraordinarily wide and damaging to Horizon’s defense.
The MDL is relatively small. With only ~273 cases, this is one of the smallest federal mass torts. A smaller case count can actually make individual settlements more valuable, since Horizon doesn’t face the existential threat of paying thousands of claimants. It also makes a global resolution much more achievable.
The timing gap is undeniable. From January 2020 (FDA approval) to July 2023 (label update) is 3.5 years. During that entire window, patients received no warnings about hearing risks, despite mounting scientific evidence. That timeline is central to every case.
Horizon’s own data may hurt them. Internal documents from Horizon’s own Study 401 reportedly showed hearing-related adverse events in up to 40% of patients. If plaintiffs can establish Horizon knew this and still didn’t warn patients, the liability picture becomes much clearer.
Do You Need a Lawyer to File a Tepezza Claim?
Quick Answer: Yes — because this is not a class action, you must have a lawyer to file a Tepezza lawsuit. There is no claim form or self-filing option. The good news is that virtually all Tepezza attorneys work on contingency, so you pay nothing upfront and nothing unless they recover money for you.
How Contingency Fee Arrangements Work
Tepezza attorneys typically charge between 33% and 40% of any recovery. If you receive a $200,000 settlement, your attorney takes $66,000–$80,000 and you receive the remainder ($120,000–$134,000). If the case is lost or dismissed, you owe nothing.
This arrangement exists because:
- Most patients can’t afford to front thousands in legal costs
- It aligns your attorney’s incentives with yours — they only get paid if you do
- It makes legal representation accessible to everyone, not just the wealthy
When to Contact an Attorney
Contact a Tepezza attorney if:
- You received Tepezza infusions at any point after its 2020 approval
- You experienced any hearing changes during or after your Tepezza treatment
- You’re unsure whether your statute of limitations has passed (an attorney will check this for free)
- You’ve already had some hearing evaluation but never pursued legal action
Free Legal Consultation
Most major firms handling Tepezza cases offer free, no-obligation consultations. You don’t need to commit to anything — just find out if you have a case. Consider reaching out to attorneys who specialize in pharmaceutical liability and have experience with MDL litigation specifically.
Contact admin@bestlawyersinunitedstates.com for attorney referrals to firms currently accepting Tepezza hearing loss cases. Red Bull Lawsuit
Frequently Asked Questions
What is the Tepezza lawsuit? Quick Answer: It’s a product liability lawsuit alleging that Tepezza manufacturer Horizon Therapeutics failed to warn patients about the drug’s risk of causing permanent hearing loss and tinnitus.
Tepezza is an FDA-approved drug for thyroid eye disease. After it launched in 2020, patients began experiencing hearing problems — including permanent hearing loss — that were not disclosed in the drug’s original label. Hearing loss warnings weren’t added until July 2023. Patients who suffered hearing damage during that window are suing Horizon Therapeutics for failure to warn.
Who is eligible to file a Tepezza lawsuit? Quick Answer: Anyone who received Tepezza infusions for thyroid eye disease and later developed hearing loss, tinnitus, or other lasting auditory problems may be eligible.
Eligibility is strongest for patients treated before July 2023 (when warnings were finally added), who have documented hearing damage, and whose symptoms appeared during or after Tepezza treatment. Contact an attorney to confirm whether your specific situation qualifies.
How much money can I get from a Tepezza lawsuit? Quick Answer: No settlement has been finalized, but attorney estimates range from $75,000 to $500,000+ depending on injury severity.
These are projections, not guarantees. Cases involving severe, permanent hearing loss — especially where patients need hearing aids or cochlear implants — are estimated on the higher end. Cases with milder or partially resolved symptoms may receive less. Final amounts depend on what happens in settlement negotiations or at trial.
Is there a Tepezza settlement yet? Quick Answer: No. As of February 2026, no global settlement has been announced, but settlement talks are actively underway and many attorneys believe a deal is close.
In October 2025, the court struck all bellwether trial deadlines — a strong signal that settlement negotiations are progressing. A status conference was held January 8, 2026. A settlement announcement could come at any time, but has not been confirmed as of this writing.
Is there a deadline to file a Tepezza lawsuit? Quick Answer: Yes — state statutes of limitations apply, typically 2–3 years from when you were injured or discovered the injury. Some people may be running out of time.
This is not like a class action where you have a single opt-in deadline. Each state sets its own filing deadline. If you received Tepezza in 2020 or 2021 and noticed hearing problems then, your window may be narrow or closed. Contact an attorney immediately to determine your specific deadline.
How do I file a Tepezza lawsuit? Quick Answer: Contact a Tepezza attorney who will evaluate your case for free and file a lawsuit on your behalf. There is no claim form.
The process: free consultation → attorney gathers your medical records → attorney files lawsuit → case joins the MDL → settlement or trial.
Do I need a lawyer? Quick Answer: Yes. This is not a class action. You cannot self-file into this litigation. You need an attorney to file a product liability lawsuit on your behalf.
The good news is virtually all Tepezza attorneys work on contingency — no upfront cost, no fee unless you win.
What documents do I need? Quick Answer: Tepezza infusion records, audiological evaluation results, and medical records documenting the onset of your hearing symptoms.
Your attorney can help obtain these records, but start pulling together: infusion dates, prescribing doctor information, any hearing evaluations you’ve had, and documentation of ongoing symptoms.
What if I don’t have records showing I received Tepezza? Quick Answer: Contact your treating physician, hospital, or infusion center — they’re required to retain medical records for years. Your pharmacy or insurance company may also have documentation.
Your attorney will help track down these records. Don’t assume you can’t file just because you don’t have paperwork in hand right now.
When will I receive payment if there’s a settlement? Quick Answer: Settlement payments in complex MDL litigation typically take 1–2 years from the time a global settlement is announced.
After a global settlement is reached, individual case valuations must be completed, plaintiffs must accept or reject their offers, and funds must be distributed. Based on comparable pharmaceutical MDL settlements, patients who resolve their claims might receive payment 1–3 years after an announcement. No one can give you a precise date because there isn’t a settlement yet.
What if I already stopped experiencing hearing problems? Quick Answer: You may still have a viable case, especially if your hearing improved only partially or if you experienced significant disruption during the period of symptoms.
Cases involving full resolution of symptoms are worth less than cases involving permanent damage, but may still have value — particularly if symptoms lasted a significant period and affected your quality of life or ability to work.
Is this a class action lawsuit? Quick Answer: No. Tepezza lawsuits are individual product liability claims consolidated into a multidistrict litigation (MDL), not a class action.
In a class action, all plaintiffs receive the same relief from a single settlement pot. In an MDL, each plaintiff maintains their own lawsuit and receives compensation based on their individual injuries. This distinction matters: you need your own attorney, not just an opt-in form.
Who is Horizon Therapeutics and are they still around? Quick Answer: Horizon Therapeutics is the manufacturer of Tepezza. The company was acquired by Amgen in 2023 for approximately $27.8 billion.
Amgen, one of the world’s largest biotech companies, is now responsible for Tepezza and potentially for any settlements arising from these lawsuits. Amgen’s deep pockets may actually be a positive development for plaintiffs — the company has the financial capacity to settle significant claims without financial distress.
What if the statute of limitations has already passed? Quick Answer: Contact an attorney anyway. “Discovery rule” exceptions may extend your filing window, and only an attorney can tell you definitively whether you’re barred.
Many states apply a “discovery rule” that starts the clock from when you knew (or reasonably should have known) that Tepezza caused your hearing problems — not from when you first had symptoms. If you only recently connected your hearing loss to Tepezza, your window may still be open.
What symptoms should prompt me to contact an attorney? Quick Answer: Any persistent hearing change after Tepezza infusions — including ringing, muffling, reduced hearing acuity, ear fullness, or balance issues.
Don’t dismiss mild symptoms as age-related or unrelated if they appeared during or after your Tepezza treatment. Get a formal audiological evaluation and consult an attorney. Many people with “minor” hearing changes have been found on testing to have measurable sensorineural hearing loss.
What happens if Horizon wins the bellwether trials? Quick Answer: Defense wins in bellwether trials would weaken plaintiffs’ leverage significantly and could result in lower settlement offers or dismissals.
Bellwether trials are specifically designed to test how juries respond to the evidence. A plaintiff win signals strong jury sympathy and increases settlement pressure on Horizon. A defense win signals the opposite. Most observers expect at least some plaintiff-favorable outcomes given the strength of the scientific evidence, but outcomes are never guaranteed.
Will a Tepezza settlement affect my taxes? Quick Answer: Compensation for physical injury and medical expenses is generally not taxable. Portions of your award for emotional distress or punitive damages may be taxable. Consult a tax professional.
The IRS generally excludes personal injury compensation from taxable income under Section 104, but there are nuances — especially for punitive damages or interest. Your attorney may be able to structure your settlement to maximize the tax-free portion.
How do I find a legitimate Tepezza attorney? Quick Answer: Look for attorneys with verified pharmaceutical injury MDL experience, who offer free consultations and work on contingency.
Red flags: attorneys who demand upfront fees, who are newly formed firms with no track record, or who make guarantees about outcomes. Good signs: existing MDL leadership positions, experience with similar drug injury cases, and clear contingency fee agreements.
Lawsuit Overview at a Glance
| Key Fact | Details |
|---|---|
| Drug | Tepezza (teprotumumab-trbw) |
| Manufacturer | Horizon Therapeutics (subsidiary of Amgen since 2023) |
| Condition treated | Thyroid eye disease (TED) |
| FDA approval | January 2020 |
| Injury alleged | Permanent hearing loss, tinnitus, Eustachian tube dysfunction |
| Warning label update | July 2023 |
| Type of litigation | Federal MDL (not class action) |
| Court | Northern District of Illinois |
| Judge | U.S. District Judge Thomas Durkin |
| Cases pending (Feb 2026) | ~273 active; 282 total filed |
| Settlement status | No settlement announced; negotiations underway |
| Estimated case values | $75,000 – $500,000+ per case |
| Attorney fees | Contingency (typically 33–40% of recovery) |
| Upfront cost to file | $0 (contingency arrangements) |

Comments