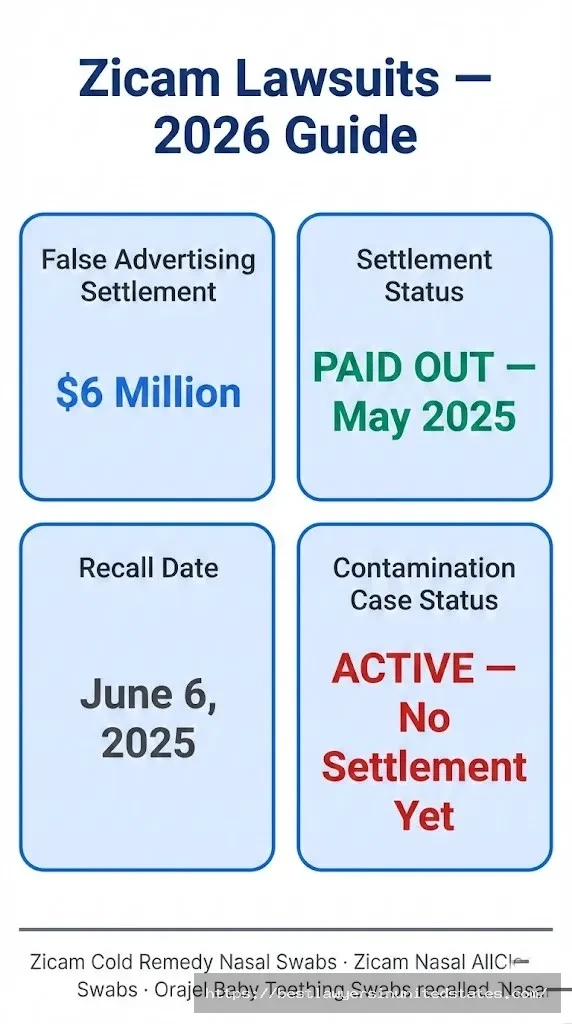
There are actually three separate Zicam lawsuits you should know about in 2026, and confusing them could cost you real money or lead you to miss important legal deadlines. The most recent — a class action filed in 2025 over fungal contamination in Zicam nasal swabs — is still active and has no settlement yet. An earlier false advertising class action settled for $6 million, with payments already distributed starting in May 2025. And dating back even further, a series of anosmia (loss of smell) lawsuits resulted in a roughly $12–15.5 million settlement over a decade ago. Facial abuse lawsuit
Quick Answer: The $6 million Zicam false advertising settlement claim deadline passed on February 21, 2025, and payments went out in May 2025. If you used Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs, or Orajel Baby Teething Swabs before the June 2025 recall and experienced illness, a newer fungal contamination class action is currently pending — no settlement has been reached yet, but attorneys are actively taking cases.
This guide covers every active and recently settled Zicam lawsuit, what happened with each, and exactly what your options are in 2026 — whether you bought Zicam for a cold, used the recalled nasal swabs, or lost your sense of smell years ago. Spectrum Lawsuit 2026
What Are the Zicam Lawsuits About?
Zicam is a brand of over-the-counter cold remedy products made with zinc. It’s sold in many forms — RapidMelts, oral mist, nasal sprays, nasal swabs, and fruit drops — at retailers like CVS, Walmart, Walgreens, Kroger, and Rite Aid. Zicam products have been available in the U.S. since 1999.
Over the years, Church & Dwight Co., Inc. (which owns the Zicam brand, manufactured originally by Matrixx Initiatives) has faced legal challenges on three separate fronts: misleading advertising claims, dangerous loss-of-smell side effects from nasal products, and most recently, life-threatening fungal contamination in recalled swab products.
Each lawsuit involves different products, different harms, and different legal claims. Understanding which one applies to you is the first step.
Lawsuit Overview Table
| Lawsuit | Issue | Defendant | Settlement Amount | Status (Feb 2026) |
|---|---|---|---|---|
| False Advertising (2022–2025) | Claimed products shorten colds without proof | Church & Dwight Co. | $6 million | CLOSED — paid out May 2025 |
| Anosmia / Loss of Smell (2009–2011) | Nasal gel caused permanent loss of smell | Matrixx Initiatives | ~$12–15.5 million | CLOSED — long resolved |
| Fungal Contamination (2025–present) | Recalled swabs contaminated with dangerous fungi | Church & Dwight Co. | Pending — no settlement yet | ACTIVE |
Lawsuit #1: The $6 Million False Advertising Settlement (Now Closed)
Background
Church & Dwight Co., the manufacturer of Zicam, agreed to settle a $6 million class action lawsuit filed in 2022. The lawsuit alleged that the company misled consumers by falsely claiming its products could shorten the duration and severity of colds and flu.
The class action lawsuit, Vance, et al. v. Church & Dwight Co. Inc., Case No. 24LA190, was filed in response to allegations that Zicam’s advertising was deceptive. Plaintiffs argued that the company’s marketing statements misled consumers into believing that Zicam products had clinically proven benefits in combating the common cold.
Zicam’s marketing centered on the idea that its zinc formula could “shorten colds” — a claim that, according to the lawsuit, wasn’t backed by solid science. Consumers paid a premium for that promise, and the case argued they deserved their money back.
What the Lawsuit Alleged
The plaintiffs laid out several specific claims against Church & Dwight:
- Zicam’s advertising created a false perception that products were more effective than the science showed
- Scientific studies cited in marketing campaigns did not conclusively support the brand’s “shortens colds” claims
- Consumers relied on deceptive advertising when purchasing Zicam products, leading to financial harm
- The amount of zinc in products was not clearly listed on labels, making it harder for consumers to evaluate the claims
Church & Dwight hasn’t admitted any wrongdoing but agreed to pay $6 million to resolve the false advertising class action. As part of the settlement, Church & Dwight also agreed to modify its Zicam labels to include qualifying statements and disclosures such as “individual results may vary.”
False Advertising Lawsuit Timeline
| Date | Event | Details |
|---|---|---|
| 2022 | Class action filed | Filed in California; Case No. 24LA190 |
| 2024 | Settlement negotiated | Church & Dwight agreed to $6M fund |
| October 17, 2024 | Purchase cutoff date | Only purchases before this date qualify |
| December 18, 2024 | Opt-out / objection deadline | Last day to exclude yourself or object |
| February 21, 2025 | Claim deadline (PASSED) | No new claims accepted after this date |
| February 28, 2025 | Final approval hearing | Court reviewed settlement terms |
| May 2025 | Payments distributed | Settlement administrator sent payments to approved claimants |
Who Qualified (For Reference)
If you purchased one or more Zicam-branded products in the United States before October 17, 2024, you may have qualified to submit a claim for a cash payment. All Zicam-branded products purchased before October 17, 2024 were included in the settlement. Claims were limited to one valid claim per household.
With proof of purchase: $5 per product, up to $30 for six products. Without proof of purchase: $2.50 per product, up to $5 for two products.
This settlement is now closed. If you submitted a claim before February 21, 2025, you should have received payment by mid-2025. If you missed the deadline, you cannot file for this settlement.
Covered Products (False Advertising Settlement)
| Product Type | Examples | Notes |
|---|---|---|
| RapidMelts | Various flavors and formulas | All versions included |
| Oral Mist | Zicam Cold Remedy Oral Mist | Included |
| Fruit Drops | Multiple varieties | Included |
| Most Zicam cold products | Purchased before Oct. 17, 2024 | Any Zicam-branded product |
Lawsuit #2: The Anosmia (Loss of Smell) Lawsuits — Historical Context
This is the oldest set of Zicam lawsuits, and it’s been resolved for years. But it’s important context because it shaped how regulators and courts view Zicam products.
What Happened
Zicam lawsuits are defective drug cases in which those who lost some of their sense of smell as a result of using the over-the-counter cold remedy Zicam sought monetary compensation from Matrixx Initiatives, Inc., the maker of Zicam. Zicam has been reported to cause anosmia (the loss of smell), which can significantly impair a person’s quality of life.
In 2009, the FDA issued a warning against using Zicam nasal sprays and gels. The agency had received at least 120 adverse event reports linking Zicam Cold Remedy Nasal Gel, Zicam Cold Remedy Nasal Swabs, and Zicam Cold Remedy Swabs Kids Size to loss of smell.
The Settlement
The makers of Zicam indicated that they were offering $15.5 million to settle lawsuits brought by consumers who alleged that they lost their sense of taste and smell due to the company’s recalled line of nasal sprays and gels. The settlement included 1,014 plaintiffs and 1,127 claimants.
This litigation resolved over a decade ago. If you used the old nasal gel products pre-2009 and suffered anosmia, that window is long closed. However, if you used recalled 2025 swab products and are experiencing smell or taste issues, that may connect to the new fungal contamination case — discussed next.
Anosmia Lawsuit Timeline
| Date | Event |
|---|---|
| 1999 | Zicam products launch in U.S. market |
| 2009 | FDA issues warning; recalls Zicam nasal gel products |
| 2009–2011 | Anosmia lawsuits filed and consolidated |
| ~2011 | Settlement reached for ~$12–15.5 million |
| Present | This litigation is fully resolved |
Lawsuit #3: The 2025 Fungal Contamination Class Action (ACTIVE)
This is the most urgent Zicam legal matter as of February 2026. If you used any of the recalled Zicam swab products — especially if you or a family member got sick — this section is the most important for you to read.
What Happened
Church & Dwight Co., Inc. issued a voluntary nationwide recall of three of their product lines on June 9, 2025. The lots in question — Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs, and Orajel Baby Teething Swabs — were found to have a microbial fungal contamination in the cotton swabs of these products that could cause life-threatening blood infections.
According to the 20-page Zicam lawsuit, the hazardous fungi Penicillium sumatraense, Penicillium citrinum and Exophiala spiniphera were found in Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs and Orajel Baby Teething Swabs made and sold by defendant Church & Dwight Co.
The recall was announced June 6, 2025 and expanded four days later on June 10. Shortly after, consumers filed a class action arguing the recall itself was inadequate. Red Bull Lawsuit
Why the Lawsuit Says the Recall Wasn’t Enough
Plaintiff Swetz claims Church & Dwight’s recall of the Zicam and Orajel products on June 6 and June 10, 2025, was insufficient since it failed to adequately inform consumers of the potential health risks, nor did it provide clear instructions for obtaining refunds.
The lawsuit contends that the company’s recall was “deliberately designed to preclude the vast majority of consumers from receiving a refund.” The filing points out that consumers who threw away recalled Zicam and Orajel swabs are not entitled to a refund — “any consumer who is made aware of the recall will be predisposed to throwing the products away,” the lawsuit states, “which would eliminate entitlement to a refund, even if the consumer maintained proof of purchase.”
Recalled Products (2025 Fungal Contamination)
| Product | UPC | Recall Scope | Risk |
|---|---|---|---|
| Zicam Cold Remedy Nasal Swabs | 732216301205 | All lots within expiry | Fungal blood infection |
| Zicam Nasal AllClear Swabs | 732216301656 | All lots within expiry | Fungal blood infection |
| Orajel Baby Teething Swabs | 310310400002 | All lots within expiry | Especially dangerous for infants |
Not recalled: Zicam RapidMelts, Zicam Oral Mist, Zicam fruit drops, and all other non-swab Zicam products remain unaffected.
Health Risks from the Contaminated Swabs
The contamination poses a significant health risk, particularly to children and individuals with compromised immune systems or underlying medical conditions. Using these contaminated swabs could lead to serious and potentially life-threatening blood infections, especially in users whose nasal mucosa may be compromised due to inflammation or mechanical injuries.
Specific health risks include:
- Fungemia — fungal blood infections requiring IV antifungal treatment
- Sepsis — life-threatening systemic response if infection goes untreated
- Pediatric risk — infants using teething swabs face especially high risk due to immature immune systems
- Altered sense of smell or taste (particularly relevant given Zicam’s history)
Who May Qualify for the Contamination Lawsuit
| Eligibility Factor | Details |
|---|---|
| Product used | Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs, or Orajel Baby Teething Swabs |
| Timing | Purchased or used before June 6, 2025 recall |
| Location | United States and Puerto Rico |
| Health impact | Experienced fever, chills, systemic infection, or blood infection symptoms after use |
| Medical confirmation | Sought treatment for possible fungal/bacterial infection |
| Proof helpful but not required | Retain product, box, lot number, or purchase receipt if possible |
Current Status (February 2026)
The fungal contamination lawsuit filed in 2025 (Swetz v. Church & Dwight Co.) is pending as of this writing. No settlement has been announced. Attorneys handling the case are actively signing up clients who used the recalled products and suffered health consequences.
Product liability claims typically must be filed within 1–3 years depending on state laws. For example: New York: 1 year, California: 3 years. Prompt action is essential to preserve medical documentation and product evidence.
If you or a family member used the recalled swabs and became ill, do not wait. The statute of limitations clock starts when you were diagnosed or became aware of your injury.
How Much Could You Get from the Contamination Lawsuit?
Because no settlement has been reached yet, there are no guaranteed payout amounts for the 2025 fungal contamination case. However, similar product contamination and defective drug cases give us a reasonable range to consider.
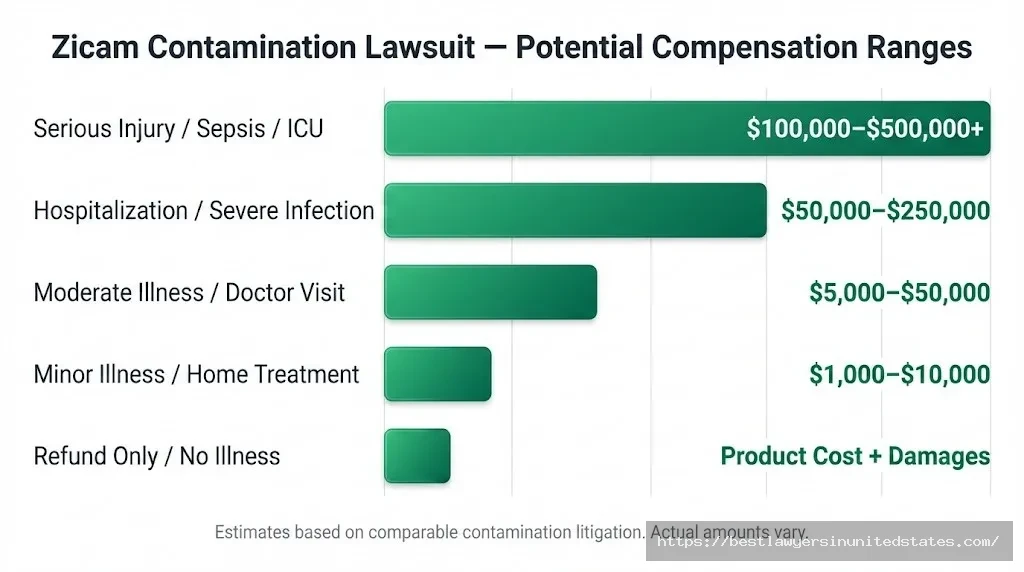
Potential Compensation by Injury Type
| Injury / Harm | Potential Compensation Range | Factors That Affect Amount |
|---|---|---|
| No illness (refund only) | Product cost + damages | Based on purchase price |
| Minor illness / treated at home | $1,000–$10,000 | Documentation, severity |
| Moderate illness / doctor visit | $5,000–$50,000 | Medical bills, lost wages |
| Hospitalization / serious infection | $50,000–$250,000+ | Length of stay, treatment |
| Life-threatening / ICU / sepsis | $100,000–$500,000+ | Long-term impact, disability |
| Pediatric / infant cases | Often higher | Vulnerability, developmental impact |
Important: These are estimates based on comparable contamination litigation. Your actual compensation will depend on the specific facts of your case, the strength of evidence, and how many claimants file claims.
What the $6 Million False Advertising Settlement Paid
For reference, the closed false advertising case paid:
| Claim Type | Payout Per Product | Maximum Payout |
|---|---|---|
| With proof of purchase | $5.00 per product | $30.00 (up to 6 products) |
| Without proof of purchase | $2.50 per product | $5.00 (up to 2 products) |
What You Should Do Right Now (2026 Action Plan)

Your next steps depend on which lawsuit applies to you.
If You Used the Recalled 2025 Swabs
Step 1: Stop using the product immediately. If you still have any Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs, or Orajel Baby Teething Swabs, do not use them. Check the UPC numbers listed above.
Step 2: Document your use. Write down when you used the product, how many times, and what symptoms you experienced afterward. Note any dates you sought medical attention.
Step 3: Keep the product if you have it. If you still have the box, lot number, or the swabs themselves, hold onto them. They may be important as evidence.
Step 4: See a doctor if you have symptoms. If you’ve had fever, chills, unusual fatigue, or any signs of infection after using the recalled products, get evaluated. Ask your doctor to document a possible fungal exposure. This medical record is critical for your case.
Step 5: Contact a product liability attorney. Because this case is still in litigation — no settlement exists yet — you need a lawyer to pursue individual compensation for serious injuries. Most personal injury attorneys handle these cases on contingency (no fee unless you win).
Step 6: Request a refund from Church & Dwight. Consumers are asked to stop using affected products immediately and contact Church & Dwight Co., Inc. for a full refund. You can contact them through their website or call their Consumer Relations Hotline at (800) 981-4710 (Mon-Fri, 9am-5pm ET).
Step 7: Report to the FDA. Any adverse reactions may be reported to the FDA’s MedWatch adverse event reporting program at fda.gov/safety/medwatch.
If You Filed a Claim Before February 21, 2025 (False Advertising)
Check whether you’ve received payment. In May 2025, the settlement administrator for the Zicam cold and virus class action settlement began issuing payments to approved claimants. If you submitted a claim and haven’t received payment, contact the settlement administrator:
- Phone: 1-833-322-3987
- Email: info@ColdVirusSettlement.com
- Mail: Zicam Products Settlement Administrator, 1650 Arch Street, Suite 2210, Philadelphia, PA 19103
If you missed the February 21, 2025 deadline, you cannot file a claim in that settlement. No exceptions.
If You Lost Your Sense of Smell After Using Zicam
If you used old Zicam nasal gel or nasal swab products before 2009 and experienced anosmia, that litigation is resolved. However, if you used the 2025 recalled swab products and are now experiencing smell or taste issues, that may be relevant to the current fungal contamination case. Speak with an attorney.
Key Deadlines & Critical Dates
| Date | Event | What It Means for You |
|---|---|---|
| February 21, 2025 | False advertising claim deadline | PASSED — cannot file |
| February 28, 2025 | Final approval hearing (false advertising) | Court approved the settlement |
| May 2025 | Payments distributed | Should have arrived by mid-2025 |
| June 6, 2025 | Zicam swab recall announced | Stop using recalled products |
| June 10, 2025 | Recall expanded | Additional lots added |
| June 2025 | Fungal contamination class action filed | Swetz v. Church & Dwight |
| 2026 (ongoing) | Fungal contamination case pending | No settlement yet; attorney sign-ups open |
| Varies by state | Statute of limitations for contamination injuries | 1–3 years from diagnosis date |
⚠️ Do not delay if you used the recalled swabs and got sick. State statutes of limitations mean your window to file a personal injury claim is limited and starts from when you knew or should have known about your injury.
Do You Need a Lawyer?
Quick Answer: For the closed false advertising settlement, you did not need a lawyer — it was designed for self-filing. For the active fungal contamination lawsuit, especially if you suffered a serious illness, an attorney is strongly recommended.
For the False Advertising Settlement (Closed)
That process was built to be handled without legal help. If you filed and got paid, you’re done. If you missed the deadline, there’s nothing a lawyer can do to reopen your claim.
For the Fungal Contamination Case (Active)
If you experienced a serious health consequence from the recalled swabs — hospitalization, significant infection, or lasting health effects — you should speak with a product liability attorney. Here’s why:
- No class action settlement has been reached, so there’s no claims portal to file through yet
- Individual injury claims (especially serious ones) are handled separately from class actions and can yield much higher compensation
- A personal injury attorney can evaluate your specific situation, gather medical evidence, and negotiate on your behalf
- Most product liability cases are taken on contingency — you pay nothing unless you win
Finding Legal Help
- Look for attorneys who specialize in product liability or defective drug cases
- Many offer free initial consultations
- Search your state bar’s referral service for verified attorneys
- You can also contact the law firm that filed the contamination class action: ClassAction.org has tracked this case from filing
How the Zicam Lawsuits Compare to Similar Cases
Understanding where the Zicam settlements fall relative to similar consumer product litigation helps set reasonable expectations.
| Lawsuit | Settlement Amount | Key Issue | Status |
|---|---|---|---|
| Zicam False Advertising (2025) | $6 million | Misleading cold remedy claims | Resolved — paid out |
| Zicam Anosmia (2011) | ~$12–15.5 million | Loss of smell from nasal gel | Resolved |
| Zicam Fungal Contamination (2025–) | Pending | Dangerous fungi in recalled swabs | Active |
| Airborne False Advertising | $23.3 million | Similar “shorten cold” claims | Resolved |
| EZShield Contaminated Product | Multiple millions | Product contamination injury | Resolved |
| OTC Drug Contamination Cases (general) | $100K–$500K+ per person | Physical injury from contamination | Varies |
The Zicam false advertising case is on the smaller end compared to similar “shorten colds” supplement cases — Airborne’s 2008 settlement, for example, was nearly four times larger. That reflects the nature of the claims and the settlement structure.
The fungal contamination case, if it leads to a class settlement, could be significantly larger depending on how many people experienced real health harm. Jon Gruden Lawsuit Against the NFL
Frequently Asked Questions
What is the Zicam lawsuit?
Quick Answer: There are actually three separate Zicam lawsuits — a false advertising case that settled for $6 million in 2025, older anosmia lawsuits that settled over a decade ago, and a new 2025 fungal contamination class action that’s still pending.
Is the Zicam settlement still open?
Quick Answer: No. The $6 million false advertising settlement claim deadline was February 21, 2025. That is closed. The 2025 fungal contamination lawsuit is active but has not yet reached a settlement.
Who qualifies for the Zicam contamination lawsuit?
Quick Answer: Anyone who purchased or used Zicam Cold Remedy Nasal Swabs, Zicam Nasal AllClear Swabs, or Orajel Baby Teething Swabs before the June 2025 recall — especially anyone who experienced illness afterward — may have a valid claim.
I missed the February 2025 claim deadline. Can I still get money?
Quick Answer: Not from the false advertising settlement — that window is closed with no exceptions. However, if you used the 2025 recalled swab products and suffered health consequences, that’s a separate, still-active case.
What were the recalled Zicam products?
Quick Answer: Zicam Cold Remedy Nasal Swabs (UPC 732216301205), Zicam Nasal AllClear Swabs (UPC 732216301656), and Orajel Baby Teething Swabs (UPC 310310400002). The recall was announced June 6, 2025 and affected all lots within expiration dates.
Are all Zicam products recalled?
Quick Answer: No. Only the three swab products listed above were recalled. Zicam RapidMelts, Oral Mist, fruit drops, and other non-swab products were not affected.
What fungi were found in the recalled Zicam swabs?
Quick Answer: Penicillium sumatraense, Penicillium citrinum, and Exophiala spiniphera — all capable of causing serious bloodstream infections, particularly in people with weakened immune systems.
How do I get a refund for the recalled swabs?
Quick Answer: Call Church & Dwight’s Consumer Relations Hotline at (800) 981-4710 (Mon–Fri, 9am–5pm ET) or visit their website to request a refund for recalled products.
My child used Orajel teething swabs from the 2025 recall. What should I do?
Quick Answer: Stop use immediately. Watch for fever, unusual irritability, or signs of infection. See a pediatrician and explain the recall. Keep any remaining product or packaging. If your child was hospitalized or seriously ill, contact a product liability attorney promptly.
I received a settlement check from Zicam in 2025. Do I need to do anything else?
Quick Answer: No. If you cashed the check from the $6 million false advertising settlement, your claim in that case is resolved. That payout may have been small (between $2.50 and $30), but it was full compensation for that specific claim.
Did Church & Dwight admit wrongdoing in the Zicam lawsuit?
Quick Answer: No. Church & Dwight denies all allegations in both the false advertising and contamination cases, but agreed to settle the advertising case to avoid prolonged litigation.
What if I threw away the recalled swab products before knowing about the recall?
The contamination class action specifically addresses this issue. The lawsuit argues that the company’s recall was “deliberately designed” to prevent most consumers from getting refunds — since people naturally throw away products after hearing about safety concerns. This is one of the central arguments in the pending case, and attorneys are taking clients even without the original product.
How long do I have to file a claim in the fungal contamination case?
Statutes of limitations vary by state — typically 1 to 3 years from the date you knew or reasonably should have known about your injury. California allows 3 years for product liability cases; New York allows 1 year for some personal injury claims. Don’t wait — consult an attorney to understand your specific deadline.
What kind of symptoms should I watch for after using recalled Zicam swabs?
Signs of a fungal blood infection include fever, chills, fatigue, sweating, muscle aches, and in serious cases, confusion or organ-related symptoms. If you used the recalled products and experience any of these, see a doctor and mention the recall.
Can I file an individual lawsuit separate from the class action?
Yes, especially if you suffered serious personal injury. Individual lawsuits often result in higher compensation than class settlements, particularly for hospitalizations or lasting health damage. A product liability attorney can advise you on whether to join the class action or pursue an individual claim.
Did anyone actually get sick from the recalled Zicam swabs?
As of the writing of early news reports, no adverse reactions had been officially linked to the contaminated products. However, comments on consumer news sites have included accounts of people reporting blood infections and illness around the time of use. The full extent of health impacts is being documented through ongoing litigation.
Where can I find the most current updates on the Zicam fungal contamination lawsuit?
Check ClassAction.org and TopClassActions.com for ongoing litigation news. For FDA recall information, visit fda.gov and search “Zicam recall.” For refunds, contact Church & Dwight directly at (800) 981-4710.
Is Zicam still being sold?
The non-swab Zicam products (RapidMelts, Oral Mist, fruit drops) were not recalled and remain on shelves. The three recalled swab products were pulled from retail. Check with your retailer or the FDA’s recall database for current status.
What changes did Zicam make to its labels after the false advertising settlement?
As part of the settlement, Church & Dwight agreed to modify its Zicam labels to include qualifying statements and disclosures such as “individual results may vary,” allowing consumers to make more informed purchasing decisions.
Summary: What You Need to Know in 2026
The Zicam legal story is ongoing. The false advertising class action is resolved and paid out. The anosmia cases are ancient history. But the fungal contamination lawsuit filed after the June 2025 recall is very much alive.
If you used the recalled swab products — for yourself or a child — and experienced health problems afterward, you have potential legal rights. Time limits apply, so getting a free attorney consultation sooner rather than later protects your options.
For refunds on the recalled products, Church & Dwight’s hotline is (800) 981-4710. For legal help with injury claims, consult a product liability attorney in your state.

Comments